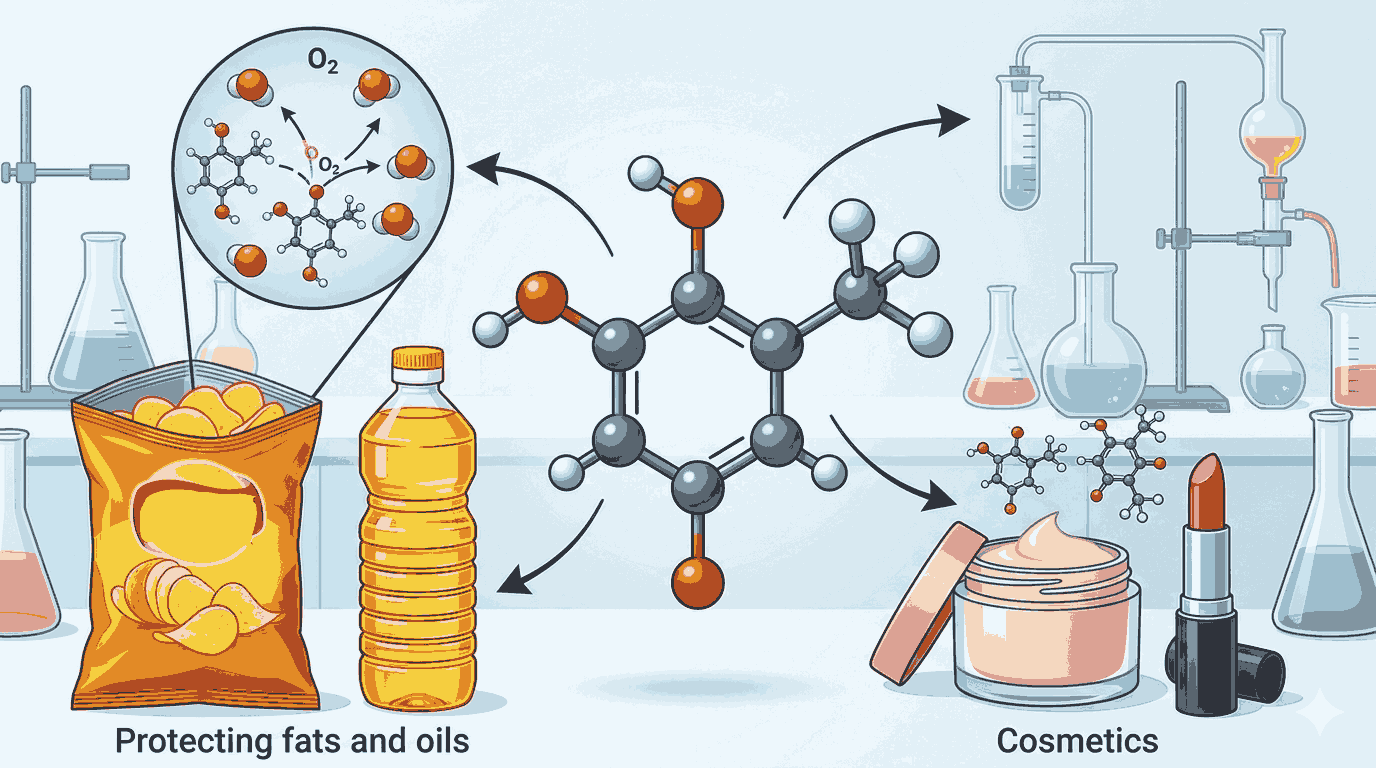
- Keneitsino Lydia
- April 14, 2026
Ultimate Revision Guide: Butylated Hydroxyanisole MCQs Class 12 Chemistry With Answers
Understanding food additives and their chemical roles is an important part of Class 12 Chemistry, especially in the chapter on Chemistry in Everyday Life. One such compound that frequently appears in exam preparation is butylated hydroxyanisole. When aspirants search for Butylated hydroxyanisole mcqs class 12 chemistry with answers, they are usually looking to strengthen their conceptual clarity about antioxidants and preservatives.
The topic of Butylated hydroxyanisole mcqs class 12 chemistry with answers is highly relevant because it connects theoretical chemistry with real-life applications. Butylated hydroxyanisole, commonly abbreviated as BHA, is a synthetic antioxidant used to prevent the oxidation of fats and oils in food. This makes it a key example when studying food preservatives in chemistry.
In many textbooks and exam guides, Butylated hydroxyanisole mcqs class 12 chemistry with answers is used to test aspirants’ understanding of antioxidants. Antioxidants like BHA work by preventing the formation of free radicals, which are responsible for the rancidity of fats. This concept is essential for students preparing for board exams and competitive exams like NEET and JEE.
When revising Butylated hydroxyanisole mcqs class 12 chemistry with answers, aspirants should focus on the chemical structure and function of BHA. It is a phenolic compound, and its antioxidant property arises from the presence of the hydroxyl group attached to the aromatic ring. This structural feature allows it to donate hydrogen atoms and stabilize free radicals.
The importance of Butylated hydroxyanisole mcqs class 12 chemistry with answers also lies in understanding its practical applications. BHA is widely used in food packaging, cosmetics, and pharmaceuticals. It helps extend the shelf life of products by preventing oxidative damage, which is a key concept in applied chemistry.
Aspirants often encounter Butylated hydroxyanisole mcqs class 12 chemistry with answers while studying preservatives and food additives. Preservatives are substances that prevent spoilage caused by microorganisms or chemical reactions. BHA falls under the category of antioxidants, which specifically prevent oxidation reactions.
Another reason why Butylated hydroxyanisole mcqs class 12 chemistry with answers is important is that it helps aspirants differentiate between various types of food additives. While preservatives prevent microbial growth, antioxidants like BHA prevent chemical degradation. This distinction is frequently tested in exams.
In the context of Butylated hydroxyanisole mcqs class 12 chemistry with answers, it is also essential to understand the concept of rancidity. Rancidity occurs when fats and oils react with oxygen, producing unpleasant odors and flavors. BHA slows down this process, making it a valuable compound in food chemistry.
The study of Butylated hydroxyanisole mcqs class 12 chemistry with answers also introduces aspirants to the broader concept of green chemistry and safe chemical usage. Although BHA is effective, its usage is regulated to ensure safety. Understanding these regulations helps students connect chemistry with health and environmental concerns.
While preparing Butylated hydroxyanisole mcqs class 12 chemistry with answers, aspirants should also compare BHA with other antioxidants like BHT (Butylated hydroxytoluene). Both compounds serve similar functions, but their chemical structures and applications may differ slightly.
The topic of Butylated hydroxyanisole mcqs class 12 chemistry with answers is often linked with questions about food safety and preservation techniques. This makes it an interdisciplinary topic, connecting chemistry with biology and environmental science.
Another important aspect of Butylated hydroxyanisole mcqs class 12 chemistry with answers is understanding how antioxidants interrupt chain reactions. Oxidation is a chain reaction process, and BHA acts as a chain-breaking antioxidant, stopping the propagation of free radicals.
When aspirants revise Butylated hydroxyanisole mcqs class 12 chemistry with answers, they should also pay attention to its solubility and stability. BHA is fat-soluble, which makes it particularly effective in oily and fatty food products.
The repeated practice of Butylated hydroxyanisole mcqs class 12 chemistry with answers helps aspirants improve their accuracy in exams. Questions on this topic are usually straightforward but require clear conceptual understanding.
In many exam-oriented books, Butylated hydroxyanisole mcqs class 12 chemistry with answers is included under the section of antioxidants and preservatives. This ensures that students can easily locate and revise this important topic.
The concept behind Butylated hydroxyanisole mcqs class 12 chemistry with answers also helps aspirants understand the role of phenolic compounds in chemistry. Phenols are known for their antioxidant properties, and BHA is a classic example.
For better preparation, aspirants should revise Butylated hydroxyanisole mcqs class 12 chemistry with answers along with related topics like artificial sweeteners, detergents, and drugs. These topics are interconnected within the Chemistry in Everyday Life chapter.
The inclusion of Butylated hydroxyanisole mcqs class 12 chemistry with answers in study material highlights its importance in exams. It is a scoring topic if understood properly.
Moreover, practicing Butylated hydroxyanisole mcqs class 12 chemistry with answers helps aspirants build confidence in answering application-based questions. These questions test not just memory but also understanding.
Butylated Hydroxyanisole MCQs Class 12 Chemistry With Answers:
1. Sodium stearate CH₃(CH₂)₁₆COO⁻Na⁺ is an anionic surfactant which forms micelles in oil. Choose the correct statement:
A) It forms spherical micelles with CH₃(CH₂)₁₆⁻ group pointing towards the center of the sphere
B) It forms non-spherical micelles with COO⁻ group pointing outward on the surface
C) It forms spherical micelles with CH₃(CH₂)₁₆⁻ group pointing outwards on the surface of sphere
D) It forms non-spherical micelles with CH₃(CH₂)₁₆⁻ group pointing towards the center
Answer: A
2. Green chemistry in day-to-day life is in the use of:
A) Chlorine for bleaching paper
B) Large amount of water for washing clothes
C) Tetrachloroethene for laundry
D) Liquefied CO₂ for dry cleaning
Answer: D
3. Which of the following is an anionic detergent?
A) Sodium lauryl sulphate
B) Cetyltrimethyl ammonium bromide
C) Glyceryl oleate
D) Sodium stearate
Answer: A
4. Which of the following compounds is not an antacid?
A) Aluminium hydroxide
B) Cimetidine
C) Phenelzine
D) Ranitidine
Answer: C
5.Match List-I with List-II
| List-I | List-II | |
|---|---|---|
| A. Chlorophyll | I. Ruthenium | |
| B. Vitamin B₁₂ | II. Platinum | |
| C. Anticancer drug | III. Cobalt | |
| D. Grubbs catalyst | IV. Magnesium |
Options:
A) A–IV, B–III, C–II, D–I
B) A–I, B–III, C–II, D–IV
C) A–I, B–II, C–III, D–IV
D) A–IV, B–II, C–III, D–I
Answer: A
6.Match List-I with List-II
| List-I | List-II | |
|---|---|---|
| A. Be (Beryllium) | I. Treatment of cancer | |
| B. Mg (Magnesium) | II. Extraction of metals | |
| C. Ca (Calcium) | III. Incendiary bombs and signals | |
| D. Ra (Radium) | IV. Windows of X-ray tubes |
Options:
A) A–IV, B–III, C–II, D–I
B) A–I, B–III, C–II, D–IV
C) A–I, B–II, C–III, D–IV
D) A–IV, B–II, C–III, D–I
Answer: A
7.Which of the following drugs is responsible for reducing the secretion of hydrochloric acid (HCl) in the stomach?
A) Aspirin
B) Cimetidine
C) Paracetamol
D) Penicillin
Answer: B
8. Which forms micelles in aqueous solution?
A) Urea
B) Dodecyl trimethyl ammonium chloride
C) Pyridinium chloride
D) Glucose
Answer: B
9. At high concentration, soap behaves as:
A) Molecular solid
B) Associated colloid
C) Macromolecular colloid
D) Lyophilic colloid
Answer: B
10. Which represents a synthetic detergent?
A) C₁₅H₃₁COOK
B) CH₃(CH₂)₆COONa
C) Given structure
D) None
Answer: C
11. Which is added to soap for antiseptic property?
A) Sodium lauryl sulphate
B) Sodium dodecyl benzene sulphonate
C) Rosin
D) Bithional
Answer: D
12. Which is known as invert soap?
A) Pentaerythritol monostearate
B) Sodium stearyl sulphate
C) Trimethyl stearyl ammonium bromide
D) Ethoxylated nonphenol
Answer: C
13. Lauryl alcohol + H₂SO₄ forms:
A) Cationic detergent
B) Anionic detergent
C) Neutral detergent
D) Antidepressant
Answer: B
14. Reaction gives yellow dye called:
A) p-Hydroxyazobenzene
B) p-Aminoazobenzene
C) p-Nitroazobenzene
D) o-Nitroazobenzene
Answer: B
15. Correct statement:
A) Saponification yields fat
B) Drying involves hydrolysis
C) Antioxidants prevent rancidity
D) Refining involves hydrogenation
Answer: C
16. Cooking is faster in pressure cooker because:
A) Food smashed
B) Water boils at higher temp
C) Constant volume
D) Less radiation
Answer: B
17. Dalda is prepared by:
A) Oxidation
B) Reduction
C) Hydrolysis
D) Distillation
Answer: B
18. Bithional in soap acts as:
A) Softener
B) Hardener
C) Dryer
D) Antiseptic
Answer: D
19. Potassium stearate is:
A) Lyophobic colloid
B) Lyophilic colloid
C) Multimolecular colloid
D) Associated colloid
Answer: D
20. Cationic detergent in hair conditioners:
A) Sodium dodecylbenzene sulphonate
B) Sodium lauryl sulphate
C) Tetraethyl ammonium chloride
D) Cetyltrimethyl ammonium bromide
Answer: D
21.Which of the following statements are correct?
- ClO₂ is used as a bleaching agent.
- OCl⁻ salts are used as disinfectants.
- OCl⁻ undergoes disproportionation in alkaline medium.
- BrO₃⁻ acts as an oxidizing agent in acidic medium.
Options:
A) 1, 2 and 3 are correct
B) 2, 3 and 4 are correct
C) 1, 2 and 4 are correct
D) All are correct
Answer: A
22. Soaps contain carbon atoms more than:
A) 10
B) 6
C) 8
D) 12
Answer: D
23. Butylated hydroxy anisole is:
A) Antioxidant
B) Cleaning agent
C) Disinfectant
D) Antihistamine
Answer: A
Conclusion on Butylated Hydroxyanisole MCQs Class 12 Chemistry With Answers
In conclusion, mastering Butylated hydroxyanisole mcqs class 12 chemistry with answers is essential for aspirants aiming for high scores in chemistry exams. It provides a clear understanding of antioxidants, food preservation, and real-world chemical applications. By revising this topic thoroughly, aspirants can enhance both their conceptual clarity and exam performance.
