- Keneitsino Lydia
- March 24, 2026
Top Hofmann Bromamide Reaction MCQs with Explanation – Powerful Practice for NEET, JEE & CUET
The Hofmann Bromamide Reaction is one of the most important name reactions in Class 12 Organic Chemistry. It is widely tested in competitive exams like NEET, JEE, and CUET because it combines both conceptual understanding and reaction mechanism clarity. To master this topic effectively, aspirants must regularly practice Hofmann Bromamide Reaction MCQs with explanation, as it helps in building a strong foundation and improving problem-solving skills.
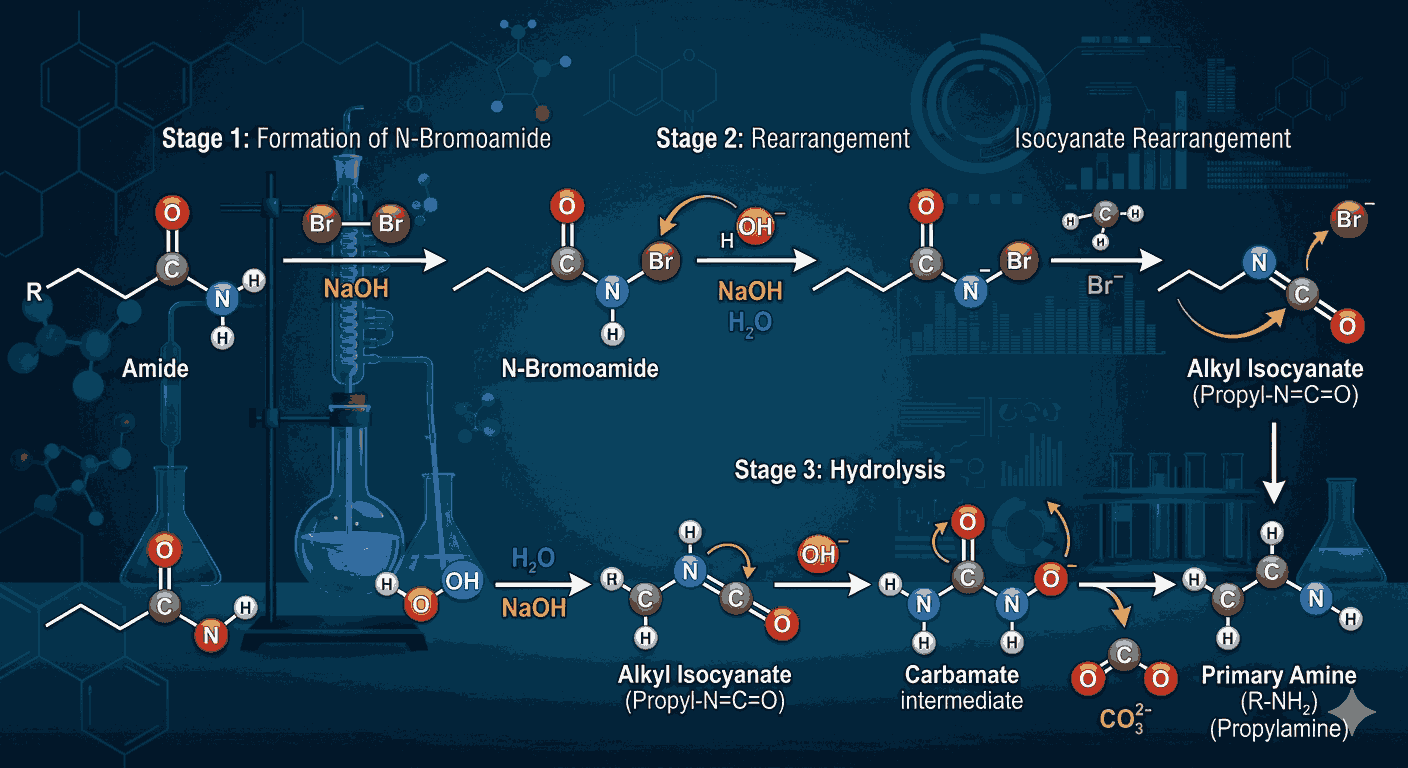
The Hofmann Bromamide Reaction involves the conversion of an amide into a primary amine with one carbon less. This reaction typically uses bromine (Br₂) and a strong base such as sodium hydroxide (NaOH). Understanding the step-by-step mechanism is crucial, and solving Hofmann Bromamide Reaction MCQs with explanation allows aspirants to visualize how the reaction proceeds through intermediates like isocyanates.
One of the key aspects of the Hofmann Bromamide Reaction is the loss of one carbon atom during the process. This makes it a very useful reaction for chain shortening in organic synthesis. By practicing Hofmann Bromamide Reaction MCQs with explanation, aspirants can easily remember this unique feature and apply it in exam-based questions.
The mechanism of this reaction includes the formation of N-bromoamide, followed by rearrangement to form an isocyanate intermediate. This intermediate then reacts with water to produce a primary amine. These steps can be confusing if studied only theoretically, but Hofmann Bromamide Reaction MCQs with explanation simplify the learning process by breaking down each step clearly.
Another important concept related to this reaction is the role of the base. Sodium hydroxide not only helps in the formation of N-bromoamide but also facilitates the rearrangement process. Practicing Hofmann Bromamide Reaction MCQs with explanation helps aspirants understand the function of each reagent involved in the reaction.
The Hofmann Bromamide Reaction is also closely related to other name reactions like Curtius and Schmidt reactions, which also involve rearrangements. By solving Hofmann Bromamide Reaction MCQs with explanation, aspirants can compare these reactions and understand their similarities and differences.
In exams, questions are often framed in the form of product prediction or mechanism-based reasoning. For example, aspirants may be asked to identify the final product after treating an amide with Br₂/NaOH. Practicing Hofmann Bromamide Reaction MCQs with explanation prepares aspirants to handle such questions quickly and accurately.
Another benefit of solving Hofmann Bromamide Reaction MCQs with explanation is that it improves speed and accuracy. With regular practice, aspirants become familiar with common patterns and can solve questions within seconds. This is particularly useful in time-bound exams where every second counts.
Moreover, Hofmann Bromamide Reaction MCQs with explanation help aspirants avoid common mistakes, such as forgetting the loss of one carbon atom or misunderstanding the reaction conditions. These MCQs provide clarity and reinforce correct concepts.
The reaction is also important in real-world applications, especially in organic synthesis where shorter chain amines are required. Understanding this practical aspect becomes easier when aspirants practice Hofmann Bromamide Reaction MCQs with explanation regularly.
Revision becomes more effective with Hofmann Bromamide Reaction MCQs with explanation, as aspirants can quickly review key concepts without going through lengthy theory. This makes it an excellent tool for last-minute preparation before exams.
Additionally, Hofmann Bromamide Reaction MCQs with explanation expose aspirants to different types of questions, including assertion-reason and case-based problems. This variety ensures comprehensive preparation for all exam formats.
Consistency in practice is essential for mastering organic chemistry. By regularly solving Hofmann Bromamide Reaction MCQs with explanation, aspirants can strengthen their understanding and improve retention of concepts.
Furthermore, Hofmann Bromamide Reaction MCQs with explanation encourage logical thinking. Aspirants learn to analyze each step of the reaction and apply their knowledge to solve complex problems.
Hofmann Bromamide Reaction MCQs with Explanation:
1. Treatment of acyl azide (RCON₃) with acidic or alkaline medium gives:
a) RCONH₂
b) R–NH₂
c) RCH₂NH₂
d) RCOCHNH
Answer: b
2. The sequence of base strength of alkyl amines is:
a) R₃N < RNH₂ > R₂NH
b) R₃N < R₂NH < RNH₂
c) R₂NH > RNH₂ > R₃N
d) RNH₂ < R₂NH < R₃N
Answer: d
3. Which method is neither for synthesis nor separation of amines?
a) Curtius reaction
b) Wurtz reaction
c) Hoffmann method
d) Hinsberg method
Answer: b
4. Identify the product:
3,4,5-tribromoaniline → (diazotisation, then H₃PO₂) → ?
a) 3,4,5-tribromobenzene
b) 1,2,3-tribromobenzene
c) 2,4,6-tribromobenzene
d) 3,4,5-tribromophenol
Answer: b
5. Electrolytic reduction of nitrobenzene in weakly acidic medium gives:
a) N-phenylhydroxylamine
b) Nitrosobenzene
c) Aniline
d) p-hydroxyaniline
Answer: c
6. Aniline + CHCl₃ + KOH gives:
a) Phenyl isocyanide
b) Phenyl cyanide
c) Phenol
d) Salicylic acid
Answer: a
7. Diethyl oxalate is used to distinguish:
a) Alcohols
b) Amines
c) Alkyl halides
d) Hydrogen in hydrocarbons
Answer: b
8. Chemicals used to manufacture methyl isocyanate (Bhopal tragedy):
a) Methylamine and phosgene
b) Phosgene and dimethylamine
c) Methylamine and phosphine
d) Phosgene and phosphine
Answer: a
9. Assertion–Reason:
Assertion: p-O₂NC₆H₄COCH₃ is prepared by Friedel-Crafts acylation of nitrobenzene.
Reason: Nitrobenzene easily undergoes electrophilic substitution.
a) Both correct, reason explains
b) Both correct, reason not explanation
c) Assertion correct, reason incorrect
d) Both incorrect
Answer: d
10. Assertion–Reason:
Assertion: Alkyl isocyanides in acidified water give alkyl formamides.
Reason: Carbon acts first as nucleophile then electrophile.
a) Both correct and reason explains
b) Both correct but not explanation
c) Assertion correct, reason incorrect
d) Both incorrect
Answer: a
11. Reagent for converting ethyl chloride to ethyl nitrite:
a) KNO₂, ethanol, water, heat
b) NaNO₂, HCl, 0°C
c) KCN, water, heat
d) AgNO₂, methanol, water
Answer: a
12. Hofmann Bromamide Reaction converts:
a) Amine→Amide
b) Amide→Amine
c)Alcohol→Amine
d) Nitrile→Amine
Answer: b
13. Decreasing order of basicity of amines:
a) (III) > (II) > (I) > (IV)
b) (II) > (III) > (I) > (IV)
c) (I) > (IV) > (III) > (II)
d) (IV) > (III) > (I) > (II)
Answer: a
14. Compound X (C₇H₇NO) gives Y with Br₂/KOH which shows mustard oil test. Identify X and Y:
a) Acetamide, ethylamine
b) Benzamide, aniline
c) Benzamide, acetanilide
d) Nitrosobenzene, aniline
Answer: b
15. Arrange amines in decreasing basicity:
a) I > III > II
b) III > I > II
c) II > I > III
d) I > II > III
Answer: b
16. Tetrahydrofuran + excess HI gives:
a) 1,4-diiodobutane
b) 1,4-butanediol
c) 2-iodotetrahydrofuran
d) 4-iodo-1-butanol
Answer: a
17. Schiff’s base is formed by reaction of aldehyde with:
a) Amine
b) Alcohol
c) Phenol
d) Carboxylic acid
Answer: a
Conclusion on Hofmann Bromamide Reaction MCQs with Explanation
In conclusion, the Hofmann Bromamide Reaction is a crucial topic that requires both conceptual clarity and practice. Hofmann Bromamide Reaction MCQs with explanation serve as an excellent resource for achieving this goal. By consistently practicing Hofmann Bromamide Reaction MCQs with explanation, aspirants can improve their understanding, enhance their accuracy, and perform exceptionally well in exams.
