- Keneitsino Lydia
- April 4, 2026
Proven Smart Prep Chloroprene and Isoprene MCQs Questions Answers to Score Higher Fast
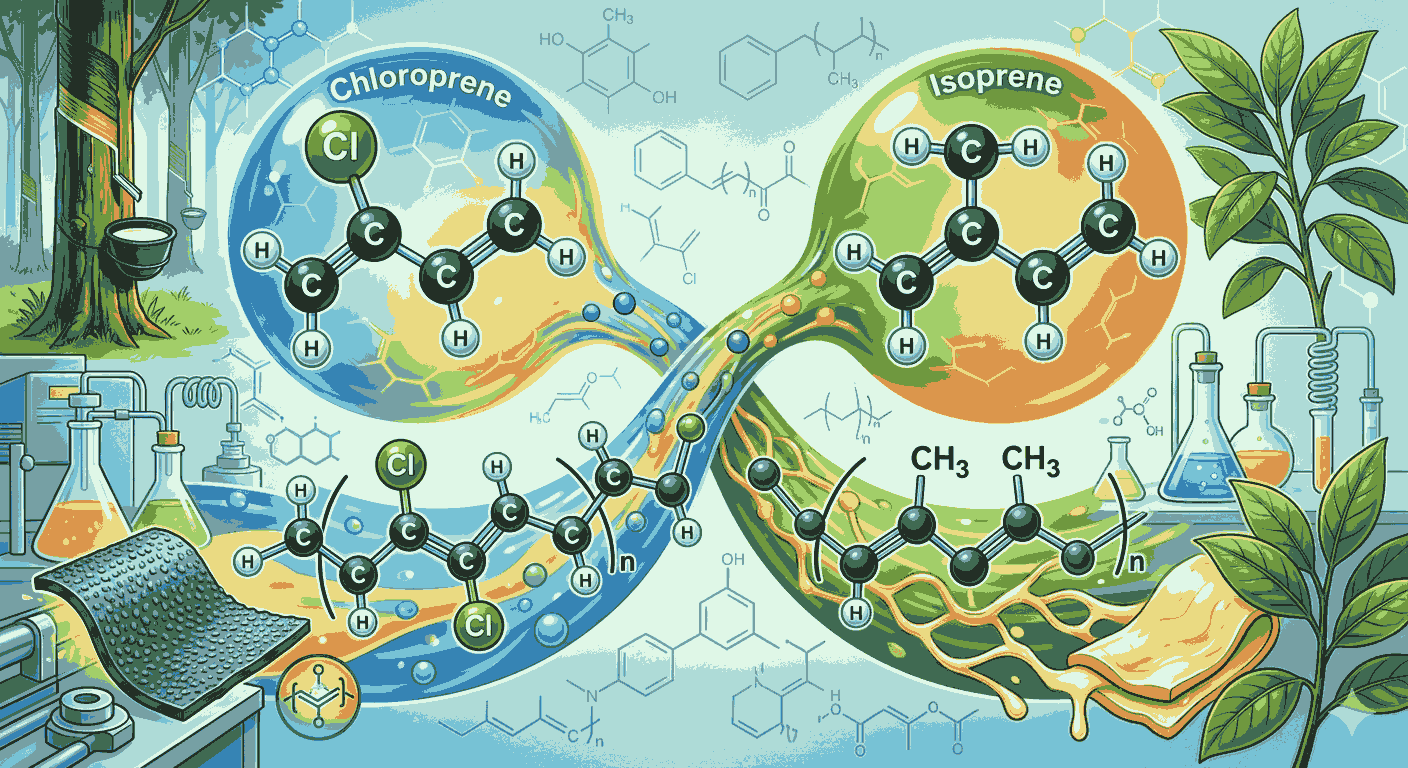
Understanding rubber chemistry becomes much easier when aspirants focus on key monomers like chloroprene and isoprene. This is why many learners actively search for Chloroprene and isoprene MCQs questions answers to strengthen their preparation for board exams and competitive tests. These monomers form the basis of important polymers, and practicing Chloroprene and isoprene MCQs questions answers helps in building a strong conceptual foundation.
Chloroprene is an important monomer used in the production of neoprene, a synthetic rubber known for its durability and resistance to chemicals. When aspirants go through Chloroprene and isoprene MCQs questions answers, they begin to understand how chloroprene undergoes addition polymerization to form neoprene. This understanding is essential for solving application-based questions in exams. Regular exposure to Chloroprene and isoprene MCQs questions answers ensures that aspirants can recall such reactions quickly.
Isoprene, on the other hand, is a naturally occurring monomer that forms natural rubber. It polymerizes to form cis-1,4-polyisoprene, which gives rubber its elasticity. By practicing Chloroprene and isoprene MCQs questions answers, aspirants can easily differentiate between natural rubber and synthetic rubber. This distinction is commonly tested in exams, making Chloroprene and isoprene MCQs questions answers highly valuable.
One of the key benefits of studying through Chloroprene and isoprene MCQs questions answers is the ability to understand polymer structures. Chloroprene contains a chlorine atom that enhances the stability of neoprene, while isoprene forms flexible chains that contribute to elasticity. Practicing Chloroprene and isoprene MCQs questions answers helps aspirants remember these structural differences effectively.
Another important aspect covered in Chloroprene and isoprene MCQs questions answers is polymerization type. Both chloroprene and isoprene undergo addition polymerization, but their resulting polymers have very different properties. By regularly solving Chloroprene and isoprene MCQs questions answers, aspirants can quickly identify reaction types and associated products.
In addition, Chloroprene and isoprene MCQs questions answers help aspirants understand real-life applications. Neoprene is used in industrial products like hoses, gaskets, and wetsuits due to its resistance to heat and oil. Natural rubber, derived from isoprene, is widely used in tyres, footwear, and elastic materials. Practicing Chloroprene and isoprene MCQs questions answers makes it easier to remember these uses during exams.
Another advantage of using Chloroprene and isoprene MCQs questions answers is improved retention of key properties. For instance, neoprene is more resistant to oxidation compared to natural rubber. Such details are often asked in objective questions, and consistent practice with Chloroprene and isoprene MCQs questions answers ensures better accuracy.
From an exam preparation perspective, Chloroprene and isoprene MCQs questions answers are extremely helpful for quick revision. Instead of going through lengthy theory, students can revise important concepts in a concise manner. This makes Chloroprene and isoprene MCQs questions answers an efficient study tool, especially before exams.
Furthermore, Chloroprene and isoprene MCQs questions answers help in improving speed and confidence. Since most entrance exams are time-bound, students need to answer quickly and accurately. Regular practice of Chloroprene and isoprene MCQs questions answers enhances both speed and decision-making ability.
Another important benefit is pattern recognition. By solving Chloroprene and isoprene MCQs questions answers, aspirants become familiar with frequently asked question types and common traps. This reduces the chances of making mistakes in exams and improves overall performance.
Additionally, Chloroprene and isoprene MCQs questions answers encourage active learning. Instead of passively reading concepts, students engage with questions that require application and analysis. This makes learning more effective and long-lasting.
Chloroprene and isoprene MCQs Questions Answers:
1. A mixture of amylose and amylopectin is called
a) Lactose
b) Starch
c) Cellulose
d) Sucrose
Answer: b
2. The condensation polymer among the following is
a) Rubber
b) Protein
c) PVC
d) Polythene
Answer: b
3. Which one of the following monomers form biodegradable polymer?
a) Urea and formaldehyde
b) Ethylene glycol and terephthalic acid
c) 3-hydroxybutanoic acid and 3-hydroxypentanoic acid
d) Phenol and caproic acid
Answer: c
4. The biodegradable polymer is
a) Buna-S
b) Nylon-6,6
c) Nylon-2-nylon-6
d) Nylon-6
Answer: c
5. Which one of the following sets forms the biodegradable polymer?
a) CH₂=CH-CN and CH₂=CH-CH=CH₂
b) H₂N-(CH₂)-COOH and H₂N-(CH₂)₅-COOH
c) All of these
d) None of these
Answer: b
6. Match the following:
a) A–II, B–III, C–I, D–IV
b) A–II, B–III, C–IV, D–I
c) A–I, B–III, C–IV, D–II
d) A–IV, B–III, C–I, D–II
Answer: a
7. Hexanedioic acid is used in the synthesis of which of the following?
a) Food preservative
b) Vinegar
c) Polymer
d) Soap
Answer: c
8. The monomers used for the synthesis of Buna-N are
a) 2-methyl-1,3-butadiene and acrylonitrile
b) 1,3-butadiene and ethynylbenzene
c) 1,2-butadiene and acrylonitrile
d) 1,3-butadiene and acrylonitrile
Answer: d
9. Which of the following polymers turns yellow on exposure to sunlight?
a) Polystyrene
b) Nylon
c) Polyethylene
d) Styrene-butadiene resin
Answer: a
10. The monomer of cellulose is
a) Fructose
b) Galactose
c) Glucose
d) None of these
Answer: c
11. Which of the following is called polyamide?
a) Rayon
b) Nylon
c) Orlon
d) Terylene
Answer: b
12. The polydispersity index (PDI) of a polymer is
a) Product of Mw and Mn
b) Sum of Mw and Mn
c) Difference of Mw and Mn
d) Ratio of Mw and Mn
Answer: d
13. Which one of the following is not an expression for molecular mass determination?
a) ΣNiMi / ΣNi
b) ΣNiMi² / ΣNiMi
c) ΣNiMi / ΣNiMi²
d) ΣCiMi / ΣCi
Answer: c
14. In the manufacture of polythene the catalyst used is
a) Fe
b) V₂O₅
c) Pt
d) TiCl₄
Answer: d
15. Natural rubber has ______ unit in its structure
a) Trans-polyisoprene
b) Cis-polyisoprene
c) Neoprene
d) Chloroprene
Answer: b
16. Hot drink cups are made up of
a) PVC
b) Polystyrene
c) Polyethylene
d) Polypropylene
Answer: b
17. 1,3-butadiene and styrene give
a) Bakelite
b) Terylene
c) Buna-S
d) Teflon
Answer: c
18. Identify the copolymer from the following
a) Given structure (Option 1)
b) –[CF₂–CF₂]ₙ–
c) Given structure (Option 3)
d) Given structure (Option 4)
Answer: a
19. If Mn = 40000 and Mw = 60000, PDI =
a) >1
b) <1
c) 1
d) 0
Answer: a
20. Which one is not used as an initiator in ionic polymerisation?
a) NaNH₂
b) SnCl₂
c) AlCl₃
d) (C₆H₅CO)₂O₂
Answer: d
21. Which polymer exhibits hydrogen bonding?
a) PVC
b) Nylon-6,6
c) Neoprene
d) Teflon
Answer: b
22. Intermolecular forces in nylon-6,6 are
a) Dipole-dipole
b) Hydrogen bonding
c) Van der Waals
d) Ionic
Answer: b
23. The monomers of Buna-S rubber are
a) Isoprene and butadiene
b) Butadiene and phenol
c) Styrene and butadiene
d) Vinyl chloride and sulphur
Answer: c
24. The monomer of neoprene is
a) 1,3-butadiene
b) 2-chloro-1,3-butadiene
c) 2-methyl-1,3-butadiene
d) Vinyl chloride
Answer: b
25. Composition of Ziegler-Natta catalyst is
a) (Et₃)₃Al·TiCl₂
b) (Me)₃Al·TiCl₂
c) (Et)₃Al·TiCl₄
d) (Et)₃Al·PtCl₄
Answer: c
26. Natural rubber is a polymer of
a) CH₂=CH–CH=CH₂
b) Isoprene
c) Given structure
d) CH₂=CH–CH₂–CH₃
Answer: b
27. Natural rubber is not used in polar regions because
a) Becomes soft below 10°C
b) Becomes brittle below 10°C
c) Melts below 10°C
d) Becomes stronger below 10°C
Answer: b
28. Cellulose diacetate fibre is
a) Natural
b) Semi-synthetic
c) Synthetic
d) None
Answer: b
29. Which statement is correct?
a) Buna-S is thermosetting
b) Neoprene is used in buckets
c) Synthesis of Buna-S needs nascent oxygen
d) Buna-S is natural polymer
Answer: c
30. Which one is not an example of chain growth polymer?
a) Neoprene
b) Buna-S
c) PMMA
d) Glyptal
Answer: d
Chloroprene and Isoprene MCQs Questions Answers
In conclusion, mastering the concepts of chloroprene and isoprene is crucial for understanding polymer chemistry. Incorporating Chloroprene and isoprene MCQs questions answers into your daily study routine can significantly enhance your preparation. Whether you are aiming for board exams or competitive tests, consistent practice with Chloroprene and isoprene MCQs questions answers will help you achieve better results and a deeper understanding of the subject.
