- Keneitsino Lydia
- April 9, 2026
Master Aspirin Structure and uses mcqs class 12 chemistry – Boost Your Score Instantly
In Class 12 chemistry, one of the most important topics under “Chemistry in Everyday Life” is aspirin. Many aspirants frequently search for Aspirin Structure and uses mcqs class 12 chemistry to strengthen their understanding before exams. This topic is not just about memorizing facts but also about understanding the structure, preparation, and applications of aspirin in real life. A clear conceptual grasp of Aspirin Structure and uses mcqs class 12 chemistry helps aspirants perform better in board exams as well as competitive exams.
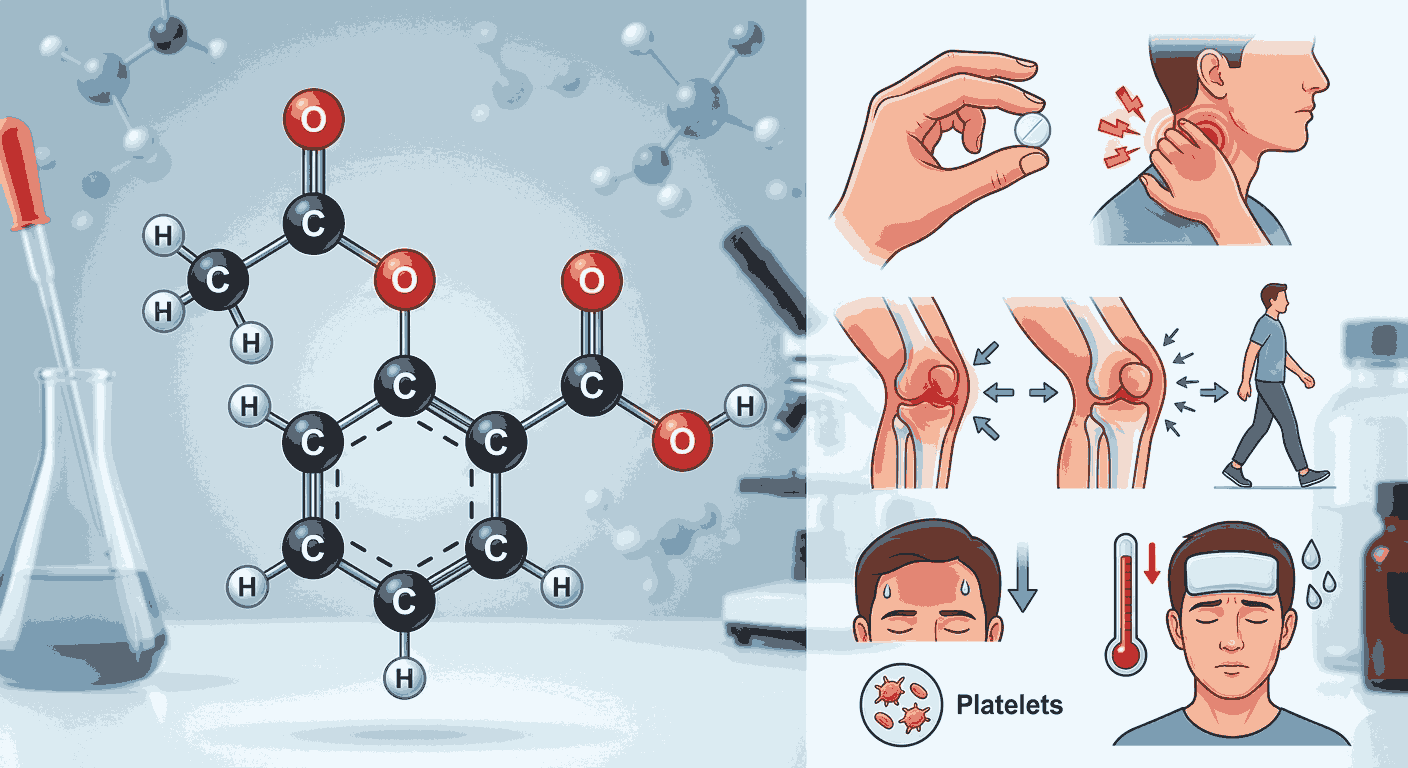
Aspirin, chemically known as acetylsalicylic acid, is a widely used drug that belongs to the class of non-steroidal anti-inflammatory drugs (NSAIDs). When studying Aspirin Structure and uses mcqs class 12 chemistry, it is important to focus on its molecular structure. The compound consists of a benzene ring attached to both an ester group and a carboxylic acid group. This dual functional group presence is often emphasized in Aspirin Structure and uses mcqs class 12 chemistry, as it plays a key role in its chemical behavior and biological activity.
Understanding the structure is the first step in mastering Aspirin Structure and uses mcqs class 12 chemistry. The ester functional group is formed by the reaction between salicylic acid and acetic anhydride. This esterification process is commonly asked in conceptual questions related to Aspirin Structure and uses mcqs class 12 chemistry, even though aspirants are not required to write full reactions in objective exams. Recognizing the functional groups and their roles makes it easier to answer structure-based questions in Aspirin Structure and uses mcqs class 12 chemistry.
When discussing the preparation of aspirin, the reaction between salicylic acid and acetic anhydride in the presence of an acid catalyst is crucial. This synthesis is a classic example often associated with Aspirin Structure and uses mcqs class 12 chemistry. The process highlights how organic compounds are modified to enhance their medicinal properties. Aspirants who understand this transformation can easily tackle application-based questions in Aspirin Structure and uses mcqs class 12 chemistry.
The uses of aspirin form another important part of Aspirin Structure and uses mcqs class 12 chemistry. Aspirin is primarily used as an analgesic, meaning it helps relieve pain. It is also an antipyretic, which means it reduces fever. Additionally, aspirin has anti-inflammatory properties, making it useful in treating conditions like arthritis. These uses are frequently tested in Aspirin Structure and uses mcqs class 12 chemistry, especially in questions that require identifying the correct function of a drug.
Another key aspect of Aspirin Structure and uses mcqs class 12 chemistry is its role as a blood thinner. Aspirin inhibits platelet aggregation, which helps prevent blood clots. This property is widely used in medical treatments to reduce the risk of heart attacks and strokes. Understanding this application provides deeper insight into the real-world importance of Aspirin Structure and uses mcqs class 12 chemistry beyond textbook knowledge.
Aspirants should also be aware of the limitations and side effects when studying Aspirin Structure and uses mcqs class 12 chemistry. Excessive use of aspirin can lead to stomach irritation because it affects the lining of the stomach. This is why aspirin is often taken with food or replaced with alternative drugs in certain cases. Such practical considerations sometimes appear in assertion-reason type questions related to Aspirin Structure and uses mcqs class 12 chemistry.
Another interesting point in Aspirin Structure and uses mcqs class 12 chemistry is the comparison with other analgesics like paracetamol. While both are used to relieve pain, aspirin has additional anti-inflammatory properties. These differences are important when solving conceptual problems in Aspirin Structure and uses mcqs class 12 chemistry, as they help distinguish between different classes of drugs.
From an exam perspective, Aspirin Structure and uses mcqs class 12 chemistry is a high-yield topic. Questions may test functional groups, preparation methods, or uses of aspirin. Therefore, repeated revision of Aspirin Structure and uses mcqs class 12 chemistry ensures that aspirants can quickly recall key information during exams. This not only improves accuracy but also saves time in competitive settings.
To effectively prepare for Aspirin Structure and uses mcqs class 12 chemistry, aspirants should focus on three main points: structure, preparation, and uses. By linking these concepts together, it becomes easier to understand how aspirin works and why it is important. This integrated approach makes Aspirin Structure and uses mcqs class 12 chemistry less about memorization and more about understanding.
Aspirin Structure and uses mcqs class 12 chemistry:
1. The medicines which prevent nausea, vomiting and motion sickness is
a) antibiotics
b) antacids
c) antispasmodics
d) all of these
Answer: c
2. A drug that commonly causes oral candidiasis is
a) sympathomimetics
b) antimuscarinic agents
c) antibiotics
d) analgesics
Answer: c
3. _______ acts both as antipyretic as well as analgesic
a) Novalgin
b) Aspirin
c) Penicillin
d) Cocaine
Answer: b
4. Which among the following has both local anaesthetic and antiseptic properties?
a) Benzyl benzoate
b) Phenol
c) Benzyl alcohol
d) n-Propyl alcohol
Answer: c
5. An antitumor drug is
a) Al(oxine)₃
b) Trans-[Pt(NH₃)₂Cl₂]
c) Cis-[Pt(NH₃)₂Cl₂]
d) K₄[Fe(CN)₆]
Answer: c
6. What are the substances which mimic the natural chemical messengers?
a) Antibiotics
b) Antagonists
c) Agonists
d) Receptors
Answer: c
7. Which of the following causes “blue baby syndrome”?
a) High concentration of lead in drinking water
b) High concentration of sulphates in drinking water
c) High concentration of nitrates in drinking water
d) High concentration of copper in drinking water
Answer: c
8. The drug used as post operative analgesia medicine is:
a) L-Dopa
b) Amoxycillin
c) Sulphadiazine
d) Morphine
Answer: d
9. Aspirin is acetyl salicylic acid, the pair of functional groups present in the compound is
a) Hydroxyl, ester
b) Carboxylic acid, hydroxyl
c) Carboxylic acid, keto
d) Carboxylic acid, ester
Answer: d
10. Identify the non-narcotic analgesic from the following
a) Diazepam
b) Ibuprofen
c) Formalin
d) Terpineol
Answer: b
11. Helium mixed with oxygen is used in the treatment of
a) Beri beri
b) Burning feet syndrome
c) Asthma
d) Diabetes
Answer: c
12. Which one of the following causes cancer?
a) 1,2-benzopyrene
b) n-Hexane
c) 2-Butene
d) Cyclohexane
Answer: a
13. Which one of the following pairs is the strongest pesticide?
a) Chloroform and benzene hexachloride
b) D.D.T. and 666
c) 666 and ether
d) Isocyanides and alcohols
Answer: b
14. Examples for bactericidal and bacteriostatic antibiotics, respectively are
a) Penicillin, ofloxacin
b) Erythromycin, tetracycline
c) Penicillin, chloramphenicol
d) Tetracycline, penicillin
Answer: c
15. Match the following columns:
<table border=”1″ cellpadding=”5″ cellspacing=”0″> <thead> <tr> <th></th> <th>List-I</th> <th></th> <th>List-II</th> </tr> </thead> <tbody> <tr> <td>A</td> <td>Oxyhaemoglobin</td> <td>I</td> <td>Analgesic</td> </tr> <tr> <td>B</td> <td>Aspirin</td> <td>II</td> <td>Oxygen carrier</td> </tr> <tr> <td>C</td> <td>Haemoglobin</td> <td>III</td> <td>Photosynthesis</td> </tr> <tr> <td>D</td> <td>Chlorophyll</td> <td>IV</td> <td>Fe²⁺ paramagnetic</td> </tr> </tbody> </table>
a) A–IV, B–I, C–II, D–III
b) A–IV, B–I, C–III, D–II
c) A–I, B–IV, C–III, D–II
d) A–I, B–IV, C–II, D–III
Answer: a
16. Parkinson’s disease is linked to abnormalities in the levels of dopamine in the body. The structure of dopamine is
a) Option 1 image
b) Option 2 image
c) Option 3 image
d) Option 4 image
Answer: c
17. Which of the following is a neurologically active drug?
a) Analgesic
b) Antacid
c) Antibiotic
d) Antihistamine
Answer: a
18. Benzene 4-hydroxy acetanilide belongs to which of the following?
a) Antipyretic
b) Antacid
c) Antiseptic
d) Antihistamine
Answer: a
19. Dettol is the mixture of
a) Chloroxylenol and bithionol
b) Chloroxylenol and terpineol
c) Phenol and iodine
d) Terpineol and bithionol
Answer: b
20. Which one of the following statements is not true?
a) Ampicillin is a natural antibiotic
b) Aspirin is both analgesic and antipyretic
c) Sulphadiazine is a synthetic antibacterial drug
d) Some disinfectants can be used as antiseptics
Answer: a
21. 2-Acetoxy benzoic acid is used as an
a) Antimalarial
b) Antidepressant
c) Antiseptic
d) Antipyretic
Answer: d
22. Which polymer is used in controlled drugs capsules?
a) PAN
b) PHBV
c) PTFE
d) SBR
Answer: b
23. Which of the given statements are correct, when carboxyhaemoglobin reaches to 3–4% in blood?
I. Leads to headache
II. Results in cardiovascular problem
III. Increases the body temperature
IV. Leads to diarrhea
a) I and II
b) I and III
c) I and IV
d) II and III
Answer: a
24. The functions of antihistamine are:
a) Antiallergic and analgesic
b) Analgesic and antacid
c) Antacid and antiallergic
d) Antiallergic and antidepressant
Answer: c
25. Which of the following compound gives pink colour on reaction with phthalic anhydride in conc. H₂SO₄ followed by treatment with NaOH?
a) Option 1 image
b) Option 2 image
c) Option 3 image
d) Option 4 image
Answer: b
26. The following compound is used as:
a) An anti-inflammatory compound
b) Analgesic
c) Hypnotic
d) Antiseptic
Answer: b
27. Assertion: Sedatives are given to patients who are mentally agitated and violent.
Reason: Sedatives are used to suppress the activities of central nervous system.
a) Both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion
b) Both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion
c) Assertion is correct but Reason is incorrect
d) Both Assertion and Reason are incorrect
Answer: a
28. Assertion: Antihistamine does not effect secretion of acid in stomach.
Reason: Antihistamine and antacids work on different receptors.
a) Both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion
b) Both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion
c) Assertion is correct but Reason is incorrect
d) Both Assertion and Reason are incorrect
Answer: a
29. Assertion: Non competitive drugs alter the shape of active site of enzyme.
Reason: They attack on the active site of enzyme.
a) Both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion
b) Both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion
c) Assertion is correct but Reason is incorrect
d) Both Assertion and Reason are incorrect
Answer: c
30. Assertion: Cimetidine is an antacid.
Reason: Antacid increases secretion of HCl from gastric cells.
a) Both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion
b) Both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion
c) Assertion is correct but Reason is incorrect
d) Both Assertion and Reason are incorrect
Answer: c
Conclusion on Aspirin Structure and uses mcqs class 12 chemistry
In conclusion, Aspirin Structure and uses mcqs class 12 chemistry is an essential topic that connects organic chemistry with real-life applications. By mastering the structure, understanding its synthesis, and remembering its uses, aspirants can confidently approach any question related to Aspirin Structure and uses mcqs class 12 chemistry. Consistent practice and conceptual clarity will ensure success in exams and build a strong foundation in medicinal chemistry.
