- Keneitsino Lydia
- March 10, 2026
Ultimate 30 Activation Energy Calculation MCQs Chemical Kinetics to Boost Your Exam Confidence
Chemical kinetics is a vital chapter in Class 12 chemistry because it explains how fast reactions occur and what factors influence reaction rates. One of the most important topics within this chapter is activation energy calculation MCQs Chemical kinetics and its role in determining the speed of a reaction. Many aspirants preparing for board exams and competitive tests regularly practice Activation Energy Calculation MCQs Chemical Kinetics to strengthen their understanding of reaction energetics and temperature dependence.
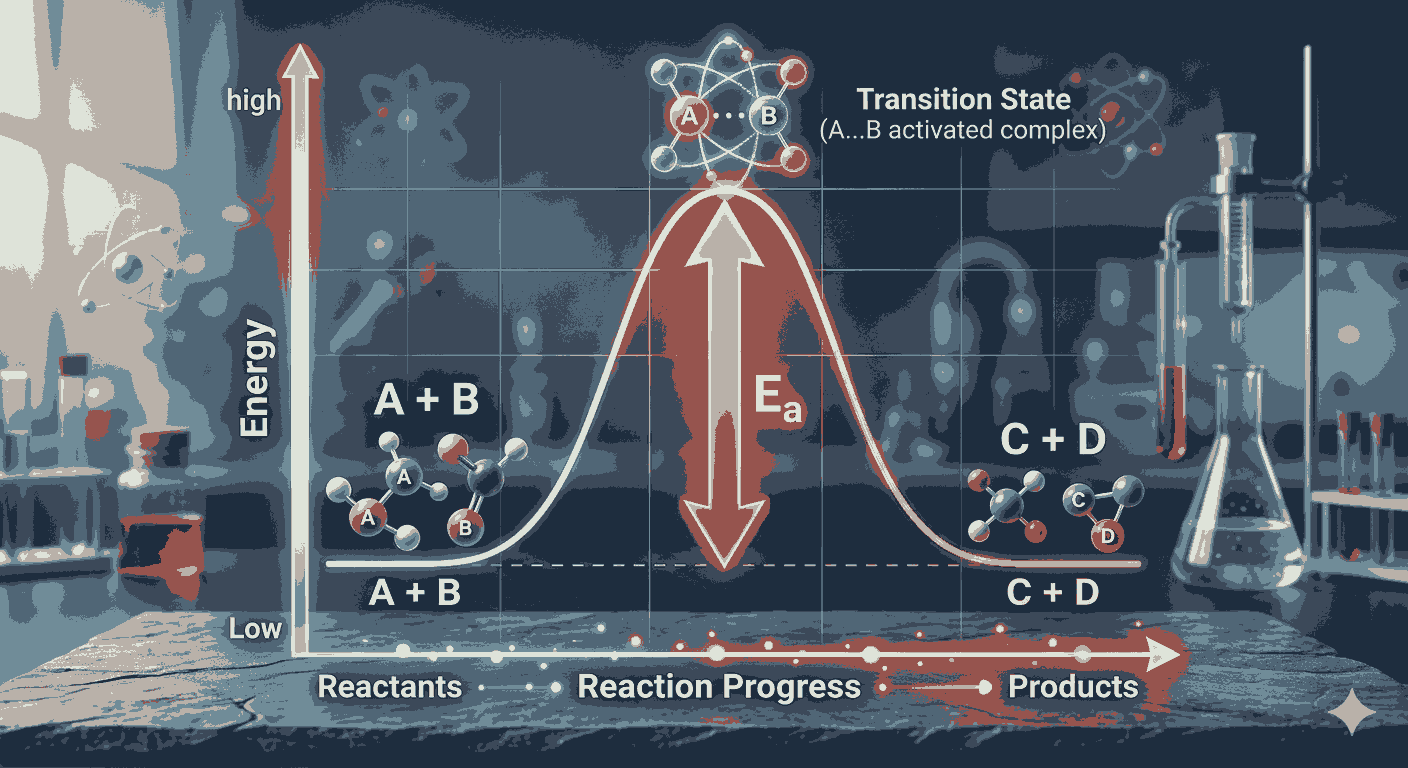
Activation energy refers to the minimum amount of energy required for reactant molecules to successfully collide and form products. Without sufficient activation energy, even frequent molecular collisions cannot lead to a chemical reaction. This concept becomes clearer when aspirants solve Activation Energy Calculation MCQs Chemical Kinetics, as these questions often focus on interpreting reaction energy profiles and understanding the energy barrier involved in chemical transformations.
The relationship between activation energy and reaction rate is explained through the Arrhenius equation. This equation expresses how the rate constant changes with temperature and activation energy. When aspirants work through Activation Energy Calculation MCQs Chemical Kinetics, they often apply the Arrhenius equation to determine activation energy or compare reaction rates at different temperatures. These activation energy calculation MCQs Chemical kinetics exercises help build strong numerical problem-solving skills in chemical kinetics.
Another key concept explored through Activation Energy Calculation MCQs Chemical Kinetics is the effect of temperature on reaction rates. As temperature increases, a larger fraction of molecules gains enough energy to overcome the activation energy barrier. This results in a higher reaction rate. By analyzing such relationships in Activation Energy Calculation MCQs Chemical Kinetics, aspirants learn how temperature influences chemical processes at the molecular level.
Graphical interpretation is also a crucial part of activation energy calculations. The Arrhenius plot, which is a graph of the logarithm of the rate constant versus the inverse of temperature, provides a convenient way to determine activation energy experimentally. Many Activation Energy Calculation MCQs Chemical Kinetics require aspirants to interpret these graphs and calculate activation energy using the slope of the Arrhenius plot. Understanding this graphical approach helps simplify otherwise complex calculations.
Catalysts also play a significant role in reactions involving activation energy. A catalyst lowers the activation energy required for a reaction, allowing more molecules to participate in the reaction at a given temperature. This concept frequently appears in Activation Energy Calculation MCQs Chemical Kinetics, where aspirants compare catalyzed and uncatalyzed reactions to understand how catalysts increase reaction rates without being consumed.
In addition to catalysts, enzymes are biological catalysts that significantly reduce activation energy in biochemical reactions. Enzymatic reactions occur much faster than ordinary chemical reactions because enzymes provide an alternative reaction pathway with lower activation energy. Such concepts are often reinforced through Activation Energy Calculation MCQs Chemical Kinetics, which help aspirants connect theoretical knowledge with biological and industrial applications.
Another important idea emphasized in Activation Energy Calculation MCQs Chemical Kinetics is the difference between activation energy and enthalpy change. While activation energy represents the energy barrier that must be overcome for a reaction to proceed, enthalpy change describes the overall energy difference between reactants and products. Understanding this distinction is essential for interpreting energy diagrams and solving kinetics problems correctly.
Practicing Activation Energy Calculation MCQs Chemical Kinetics also improves analytical thinking and exam readiness. Many competitive exams such as NEET and JEE include numerical problems based on activation energy calculations. Aspirants who regularly review Activation Energy Calculation MCQs Chemical Kinetics become more confident in applying formulas, interpreting graphs, and solving temperature-dependent reaction problems.
Activation Energy Calculation MCQs Chemical Kinetics:
1. Pseudo-First Order Reaction
Q1. Acid catalyzed hydrolysis of ethyl acetate follows pseudo-first order kinetics with respect to ester. If the reaction is carried out with large excess of ester, the order with respect to ester will be:
A. 1.5
B. Zero
C. 0.5
D. 1
Answer: B
2. Rate and pH Change
Q2. The rate of a certain reaction is given by:
Rate = k[H⁺]ⁿ.
If the rate increases 100 times when pH changes from 3 to 1, the order (n) is:
A. 2
B. Zero
C. 1
D. 1.5
Answer: C
3. Arrhenius Plot
Q3. Activation energy of a reaction can be calculated by plotting:
A. log k vs 1/T
B. log k vs 1/log T
C. k vs T
D. k vs 1/log T
Answer: A
4. Activation Energy
Q4. Which factor mainly determines the activation energy of a reaction?
A. Nature of Reactants
B.Pressure
C. Concentration
D. Volume of container
Answer: A
5. Arrhenius Relation
Q5. According to Arrhenius equation, the relationship between rate constants and temperature is:
A. ln(k2/k1)=EaR(1/T1−1/T2)ln(k_2/k_1) = \frac{E_a}{R}(1/T_1 – 1/T_2)
B. ln(k2/k1)=−EaR(1/T1−1/T2)ln(k_2/k_1) = -\frac{E_a}{R}(1/T_1 – 1/T_2)
C. ln(k2/k1)=EaR(1/T1+1/T2)ln(k_2/k_1) = \frac{E_a}{R}(1/T_1 + 1/T_2)
D. ln(k2/k1)=−EaR(1/T1+1/T2)ln(k_2/k_1) = -\frac{E_a}{R}(1/T_1 + 1/T_2)
Answer: A
6. Meaning of Activation Energy
Q6. In the Arrhenius equation k=Ae−Ea/RTk = Ae^{-E_a/RT}, EaE_a represents:
A. Energy above which not all molecules react
B. Energy below which colliding molecules will not react
C. Total energy of molecules
D. Fraction of molecules with energy above activation energy
Answer: B
7. Role of Enzymes
Q7. Enzymes increase the rate of reactions by:
A. Lowering activation energy
B. Increasing activation energy
C. Changing equilibrium constant
D. Increasing concentration
Answer: A
8. Activation Energy Calculation
Q8. Rate constant at 200 K is 10 times less than at 400 K. Activation energy is:
A. 184.2 R
B. 921.2 R
C. 460.6 R
D. 230.3 R
Answer: B
9. Effect of Catalyst
Q9. A catalyst changes the:
A. Activation energy
B. Heat of reaction
C. Final products
D. Concentration of products
Answer: A
10. Reverse Reaction Activation Energy
Q10. If forward activation energy is EaE_a, reverse activation energy:
A. Always less
B. Can be less or more
C. Always double
D. Negative
Answer: B
11. Correct Statement
Q11. In k=Ae−Ea/RTk = Ae^{-E_a/RT}:
A. k is equilibrium constant
B. A is adsorption factor
C. EaE_a is activation energy
D. R is Rydberg constant
Answer: C
12. Temperature Effect
Q12. In a zero-order reaction, rate doubles every 10°C rise. From 10°C to 100°C increase:
A. 256 times
B. 512 times
C. 128 times
D. 1024 times
Answer: B
13. Fraction of Molecules
Q13. For Ea=80.9E_a = 80.9 kJ/mol at 700 K, fraction of molecules is e−xe^{-x}. Value of x:
A. 14
B. 16
C. 7
D. 8
Answer: A
14. Arrhenius Graph Question
Q14. Temperature at which rate constant becomes 10−410^{-4} s⁻¹ is:
A. 526.3 K
B. 500.3 K
C. 520.3 K
D. 556.3 K
Answer: A
15. Formic Acid Decomposition
Q15. Rate constant at 200 K equals:
A. 10 × 10⁻⁵ s⁻¹
B. 100 × 10⁻⁵ s⁻¹
C. 12 × 10⁻⁵ s⁻¹
D. 11 × 10⁻⁵ s⁻¹
Answer: A
16. Activation Energy Calculation
Q16. Activation energy when rate increases five times from 27°C to 52°C:
A. 52 kJ/mol
B. 55 kJ/mol
C. 62 kJ/mol
D. 65 kJ/mol
Answer: A
17. Reverse Activation Energy
Q17. For exothermic reaction with Ea=30E_a = 30 kJ/mol and ΔE = −20 kJ/mol, reverse activation energy:
A. 10 kJ/mol
B. 30 kJ/mol
C. 50 kJ/mol
D. 70 kJ/mol
Answer: C
18. Potential Energy Diagram
Q18. Correct diagram corresponds to:
A. Diagram 1
B. Diagram 2
C. Diagram 3
D. Diagram 4
Answer: C
19. Arrhenius Slope
Q19. If slope of Arrhenius plot is −5×10³ K, activation energy is:
A. 83.0 kJ/mol
B. 41.5 kJ/mol
C. 83 J/mol
D. 166 kJ/mol
Answer: B
20. Activation Energy Zero
Q20. If Ea=0E_a = 0, ratio of rate constants at 273 K and 383 K is:
A. Zero
B. 1
C. 100
D. 10
Answer: D
21. Catalyst Effect
Q21. Activation energy with catalyst becomes:
A. 198 kJ/mol
B. 75 kJ/mol
C. 108 kJ/mol
D. 50 kJ/mol
Answer: B
22. Milk Spoilage
Q22. Activation energy is approximately:
A. 3.98 kJ/mol
B. 19.9 kJ/mol
C. 39.8 kJ/mol
D. 79.6 kJ/mol
Answer: A
23. Activation Energy from Graph
Q23. Activation energy equals:
A. 2/R
B. 1/R
C. R
D. 2R
Answer: D
24. Temperature Change Effect
Q24. Activation energy when rate decreases 3.555 times:
A. 100 kJ/mol
B. 50 kJ/mol
C. 25 kJ/mol
D. 10 kJ/mol
Answer: A
25. Enzyme Effect
Q25. Change in activation energy when enzyme increases rate 10610^6 times:
A. 6(2.303)RT6(2.303)RT
B. −6(2.303)RT-6(2.303)RT
C. 6RT6RT
D. −6RT-6RT
Answer: B
26. Arrhenius Equation Factor
Q26. To speed up reaction, which factor must decrease?
A. EaE_a
B. Z
C. Z and T
D. T
Answer: A
27. Reverse Activation Energy
Q27. For endothermic reaction with Ea=13E_a = 13 kJ/mol and ΔH = 2 kJ/mol, reverse activation energy:
A. 15 kJ/mol
B. 11 kJ/mol
C. 26 kJ/mol
D. −15 kJ/mol
Answer: B
28. Zero Activation Energy
Q28. If activation energy is zero, rate constant:
A. Increases with temperature
B. Decreases with temperature
C. Decreases with decreasing temperature
D. Independent of temperature
Answer: D
29. Correct Equation
Q29. Correct equation is:
A. lnk−lnA=Ea/RTlnk – lnA = Ea/RT
B. k=Ea/RTk = Ea/RT
C. lnk+lnA=Ea/RTlnk + lnA = Ea/RT
D. Ea/RT=lnA−lnkEa/RT = lnA – lnk
Answer: D
30. Activation Energy from Rate Change
Q30. If rate doubles from 300 K to 310 K, activation energy is:
A. 53.6 kJ/mol
B. 48.6 kJ/mol
C. 58.3 kJ/mol
D. 60.5 kJ/mol
Answer: A
Conclusion on Activation Energy Calculation MCQs Chemical Kinetics
In conclusion, activation energy calculation MCQs Chemical Kinetics is a fundamental concept that explains why some reactions occur rapidly while others proceed slowly. By consistently practicing Activation Energy Calculation MCQs Chemical Kinetics, aspirants gain a deeper understanding of reaction mechanisms, temperature effects, and catalytic processes. Mastering Activation Energy Calculation MCQs Chemical Kinetics ultimately helps build a strong foundation in chemical kinetics and prepares aspirants for success in both academic examinations and competitive entrance tests.
