- Keneitsino Lydia
- March 4, 2026
Zero Order and First Order Reaction MCQs Class 12: A Powerful Success Guide
Zero Order and First Order Reaction MCQs Class 12 form the backbone of chemical kinetics in Class 12 chemistry. When aspirants prepare through these MCQS , they develop conceptual clarity about rate laws, half-life equations, integrated rate expressions, and graphical interpretations. Understanding these two reaction orders is essential because many competitive exams repeatedly test these concepts in numerical and theoretical form.
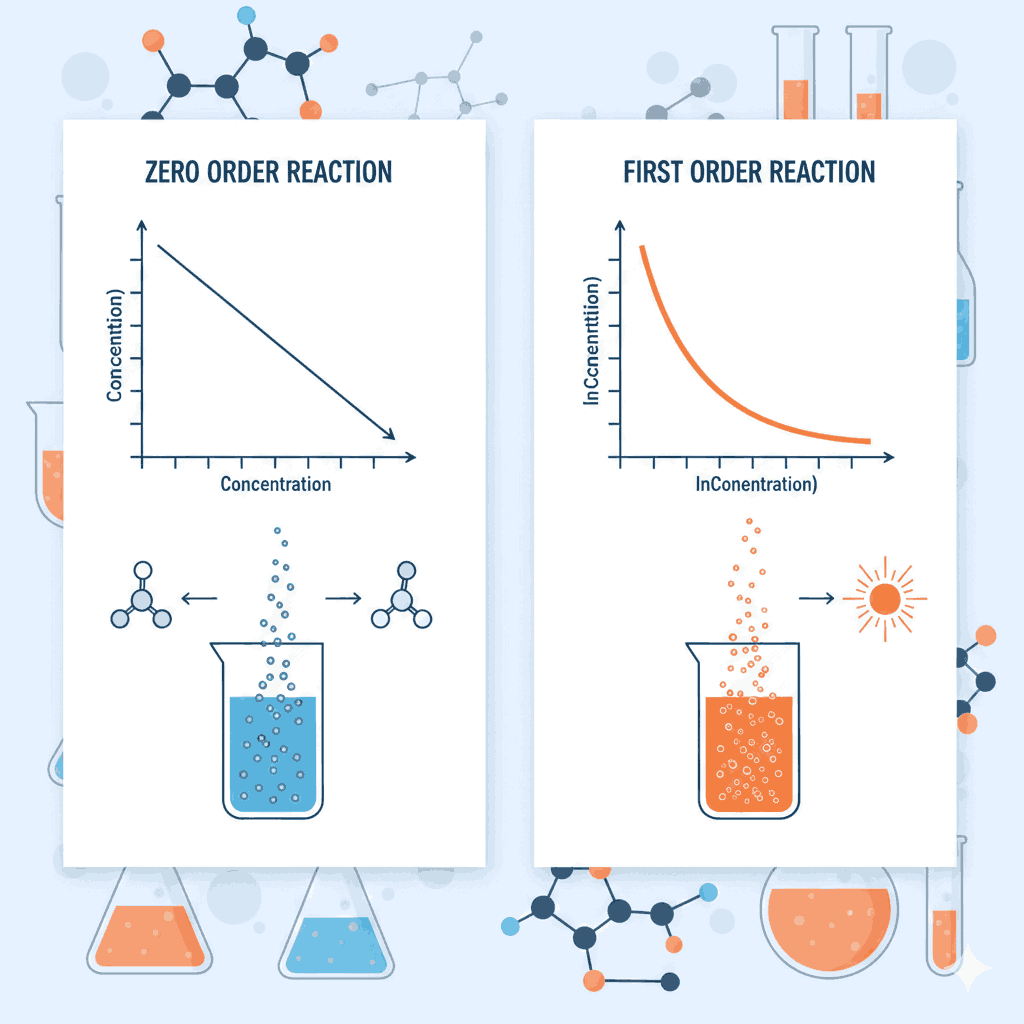
In zero order reactions, the rate of reaction is independent of the concentration of reactants. This means the rate remains constant throughout the reaction as long as external conditions such as temperature remain unchanged. In Zero Order and First Order Reaction MCQs Class 12, students frequently encounter questions related to the linear decrease of concentration with time. The integrated rate law for a zero order reaction is:
[A]=[A]0−kt[A] = [A]_0 – kt
This equation shows that concentration decreases linearly with time. Many Zero Order and First Order Reaction MCQs Class 12 also test understanding of graphical representation, where a plot of concentration versus time gives a straight line with slope equal to −k.
Another important feature emphasized in Zero Order and First Order Reaction MCQs Class 12 is the unit of the rate constant. For zero order reactions, the unit of k is mol L⁻¹ s⁻¹, which is the same as the unit of rate. This is a common conceptual trap in Zero Order and First Order Reaction MCQs Class 12, where aspirants confuse units across different orders.
First order reactions, on the other hand, depend linearly on the concentration of a single reactant. The rate law is expressed as:
Rate=k[A]\text{Rate} = k[A]
In Zero Order and First Order Reaction MCQs Class 12, one of the most frequently tested equations is the integrated rate law for first order reactions:
ln[A]=ln[A]0−kt\ln [A] = \ln [A]_0 – kt
Or in logarithmic form:
k=2.303tlog[A]0[A]k = \frac{2.303}{t} \log \frac{[A]_0}{[A]}
Mastering these formulas is essential for solving numericals appearing in Zero Order and First Order Reaction MCQs Class 12.
One of the most powerful tools in Zero Order and First Order Reaction MCQs Class 12 preparation is understanding half-life expressions. For zero order reactions, half-life depends on initial concentration:
t1/2=[A]02kt_{1/2} = \frac{[A]_0}{2k}
But for first order reactions:
t1/2=0.693kt_{1/2} = \frac{0.693}{k}
In Zero Order and First Order Reaction MCQs Class 12, this difference is repeatedly examined because it clearly distinguishes concentration dependence.
Graphical interpretation is another crucial area in Zero Order and First Order Reaction MCQs Class 12. For first order reactions, a plot of ln[A] versus time gives a straight line. In contrast, concentration versus time gives a straight line for zero order reactions. Recognizing these graphs quickly is vital for scoring well in Zero Order and First Order Reaction MCQs Class 12.
Another commonly tested idea in Zero Order and First Order Reaction MCQs Class 12 is the constancy of half-life. In first order reactions, half-life remains constant irrespective of initial concentration. However, in zero order reactions, half-life changes with concentration. This conceptual distinction is often asked indirectly in Zero Order and First Order Reaction MCQs Class 12.
Temperature effects are equally important. Although both zero and first order reactions follow the Arrhenius equation, in Zero Order and First Order Reaction MCQs Class 12, aspirants are usually asked how rate constant changes with temperature rather than concentration.
Applications of first order kinetics include radioactive decay and decomposition reactions. Many Zero Order and First Order Reaction MCQs Class 12 use radioactive decay as a standard example because it perfectly follows first order behavior. Zero order reactions are often seen in photochemical reactions or reactions occurring on catalytic surfaces, which are also commonly discussed in Zero Order and First Order Reaction MCQs Class 12.
Problem-solving strategies in Zero Order and First Order Reaction MCQs Class 12 include identifying the order from units of rate constant, checking half-life behavior, analyzing graphical data, and applying correct integrated equations.
Zero Order and First Order Reaction MCQs Class 12:
1.A chemical reaction was carried out at 300 K and 280 K. The rate constants were found to be k₁ and k₂ respectively. Then:
A. k₂ = 4k₁
B. k₂ = 0.25k₁
C. k₂ = k₁
D. k₂ = 2k₁
Answer: B
2.In the reversible reaction:
2NO2⇌k2k1N2O42NO_2 \overset{k_1}{\underset{k_2}{\rightleftharpoons}} N_2O_4
The rate of disappearance of NO₂ is equal to:
A. 2k1k2[NO2]2\frac{2k_1}{k_2}[NO_2]^2
B. 2k1[NO2]2−2k2[N2O4]2k_1[NO_2]^2 – 2k_2[N_2O_4]
C. 2k1[NO2]2−k2[N2O4]2k_1[NO_2]^2 – k_2[N_2O_4]
D. (2k1−k2)[NO2](2k_1 – k_2)[NO_2]
Answer: B
3.90% of a first order reaction is completed in 70 minutes. The velocity constant of the reaction is:
A. 0.0329
B. 0.693
C. 3.29
D. 0.0239
Answer: A
4.At 300 K, activation energy of A is higher than B by 5.75 kJ mol⁻¹ in presence of catalyst. Calculate kAkB\frac{k_A}{k_B}:
A. 1
B. 10
C. 1000
D. 100
Answer: B
5.Rate of two reactions whose rate constants are k₁ and k₂ are equal at 300 K such that:
Ea2−Ea1=2RTE_{a2} – E_{a1} = 2RT
Calculate:
lnA2A1\ln \frac{A_2}{A_1}
A. ln 4
B. 2
C. log 2
D. 2 ln 4
Answer: B
6.The rate constant, activation energy, and Arrhenius parameter of a chemical reaction at 25°C are 3.0 × 10⁻⁴ s⁻¹, 104.4 kJ mol⁻¹ and 6.0 × 10¹⁴ s⁻¹ respectively. The value of the rate constant at T → ∞ is:
A. 2.0 × 10¹⁸ s⁻¹
B. 6.0 × 10¹⁴ s⁻¹
C. 3.6 × 10³⁰ s⁻¹
D. Infinity
Answer: B
7.Two reactions R₁ and R₂ have identical pre-exponential factors. Activation energy of R₁ exceeds that of R₂ by 10 kJ mol⁻¹. If k₁ and k₂ are rate constants at 300 K, then:
lnk2k1=\ln \frac{k_2}{k_1} =
A. 8
B. 12
C. 6
D. 4
Answer: D
8.For the reaction:
2O₃ ⇌ 3O₂
Mechanism:
O₃ ⇌ O₂ + O (fast)
O + O₃ → 2O₂ (slow)
The rate law expression is:
A. r = k'[O₃]²
B. r = k'[O₃][O₂]
C. r = k'[O₃]²[O₂]⁻¹
D. Unpredictable
Answer: C
9.For reaction A₂ + B → Products with mechanism:
A₂ ⇌ 2A (fast)
A + B → Products (slow)
Rate law is:
A. rate = k[A₂][B]
B. rate = k[A₂][B]²
C. rate = k[A₂]¹ᐟ²[B]
D. rate = k[A₂]²[B]
Answer: C
10.
For NO₂(g) + CO(g) → NO(g) + CO₂(g), rate = k[NO₂]². If 0.1 mole CO is added at constant temperature, then:
A. Both k and rate remain same
B. Both increase
C. Both decrease
D. Only k increases
Answer: A
11.
For reaction Cl₂ + H₂S → S + 2H⁺ + 2Cl⁻, rate = k[Cl₂][H₂S]. Which mechanism is consistent?
A. B only
B. Both A and B
C. Neither A nor B
D. A only
Answer: D
12.
Reaction 3ClO⁻ → ClO₃⁻ + 2Cl⁻ occurs via slow first step. Rate is:
A. k₁[ClO⁻]²
B. k₁[ClO⁻]
C. k₂[ClO₂⁻][ClO⁻]
D. k₂[ClO⁻]²
Answer: A
13.
For 2N₂O₅ → 4NO₂ + O₂, if k = 3 × 10⁻⁵ s⁻¹ and rate = 2.4 × 10⁻⁵ mol L⁻¹ s⁻¹, concentration of N₂O₅ is:
A. 0.4
B. 0.8
C. 0.04
D. 0.06
Answer: B
14.
Two reactions have same k at 25°C but different at 35°C because of:
A. Effective collisions
B. Different activation energies
C. Different threshold energies
D. Higher population
Answer: B
15.
Assertion: If activation energy is zero, temperature has no effect.
Reason: Lower activation energy, faster reaction.
A. Both correct and reason explains
B. Both correct but reason not explanation
C. Assertion correct, reason wrong
D. Both wrong
Answer: B
16.
If activation energy is zero, rate constant is:
A. Increases with T
B. Decreases with T
C. Decreases with decrease of T
D. Independent of T
Answer: D
17.
Unit of rate constant same as rate for:
A. Zero order
B. First order
C. Second order
D. Third order
Answer: A
18.
For reaction A → B, k = 0.6 × 10⁻³ mol L⁻¹ s⁻¹, [A] = 5M, after 20 min concentration of B is:
A. 3.0 M
B. 0.36 M
C. 0.72 M
D. 1.08 M
Answer: C
19.
For A + B → C, R = k[A][B]². If both doubled, rate becomes:
A. Eight times
B. Double
C. Quadruple
D. Triple
Answer: A
20.
k at 27°C = 10⁻³ min⁻¹, temperature coefficient = 2. k at 17°C is:
A. 2 × 10⁻⁴
B. 5 × 10⁻²
C. 5 × 10⁻⁴
D. 10⁻²
Answer: C
21.
Quantity K in rate expression:
A. Independent of concentration
B. Arrhenius constant
C. Dimensionless
D. Independent of temperature
Answer: A
22.
For A ⇌ B (fast), A + B → C (slow), rate law is proportional to:
A. [A][B]
B. [A]²
C. [A][B]²
D. [A]²[B]
Answer: A
23.
Half-life of first order reaction = 10 min. Rate constant is:
A. 6.93 × 10⁻³ min⁻¹
B. 0.693 × 10⁻² min⁻¹
C. 6.932 × 10⁻² min⁻¹
D. 6.932 × 10⁻² min
Answer: C
24.
Unit of rate constant for zero order reaction:
A. mol L⁻¹ s⁻¹
B. L mol⁻¹ s⁻¹
C. L mol⁻² s⁻¹
D. s⁻¹
Answer: A
25.
Reaction at 320 K and 300 K with rate constants k₁ and k₂ respectively. Then:
A. k₂ = 4k₁
B. k₂ = 2k₁
C. k₂ = 0.25k₁
D. k₂ = 0.5k₁
Answer: C
26.
Half-life = 1 min 40 sec. Rate constant is:
A. 6.93 × 10⁻³ min⁻¹
B. 6.93 × 10⁻³ s⁻¹
C. 6.93 × 10⁻³ s
D. 0.693 × 10⁻³ s⁻¹
Answer: B
27.
Slope of straight line between ln k and 1/T is:
A. −Ea / 2.303R
B. −Ea / R
C. Ea / R
D. R / Ea
Answer: B
28.
Time for 90% completion = t min. Time for 99% completion (in sec) is:
A. 2t
B. t/30
C. 120t
D. 60t
Answer: A
29.
Temperature coefficient = 2.5. If k₁ = 2.5 × 10⁻³ s⁻¹, then k₂ is:
A. 1.0 × 10⁻²
B. 6.25 × 10⁻²
C. 1 × 10²
D. 6.25 × 10⁻³
Answer: D
30.
The rate constant for reaction 2N₂O₅ → 4NO₂ + O₂ is 3.0 × 10⁻⁴ s⁻¹. If rate is 2.4 × 10⁻⁵ mol L⁻¹ s⁻¹, concentration of N₂O₅ is:
A. 4
B. 1.2
C. 0.02
D. 0.08
Answer: D
Conclusion on Zero Order and First Order Reaction MCQs Class 12
Ultimately, mastering concepts behind Zero Order and First Order Reaction MCQs Class 12 strengthens both numerical solving ability and conceptual understanding. Aspirants who thoroughly practice Zero Order and First Order Reaction MCQs Class 12 develop strong analytical skills required for board exams and competitive exams alike. Consistent revision of formulas, graphical interpretations, and unit analysis ensures high accuracy while solving Zero Order and First Order Reaction MCQs Class 12, making this topic one of the most scoring areas in Class 12 chemistry.
