- Keneitsino Lydia
- April 3, 2026
Master Vulcanization and Rubber Chemistry MCQs with Answers – Boost Your Exam Score Instantly
Understanding polymers is an essential part of chemistry preparation, and Vulcanization and rubber chemistry MCQs with answers play a vital role in strengthening this concept for competitive exams. Rubber chemistry focuses on natural and synthetic rubber, their structures, properties, and modifications like vulcanization, which significantly enhances their usefulness.
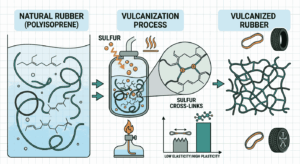
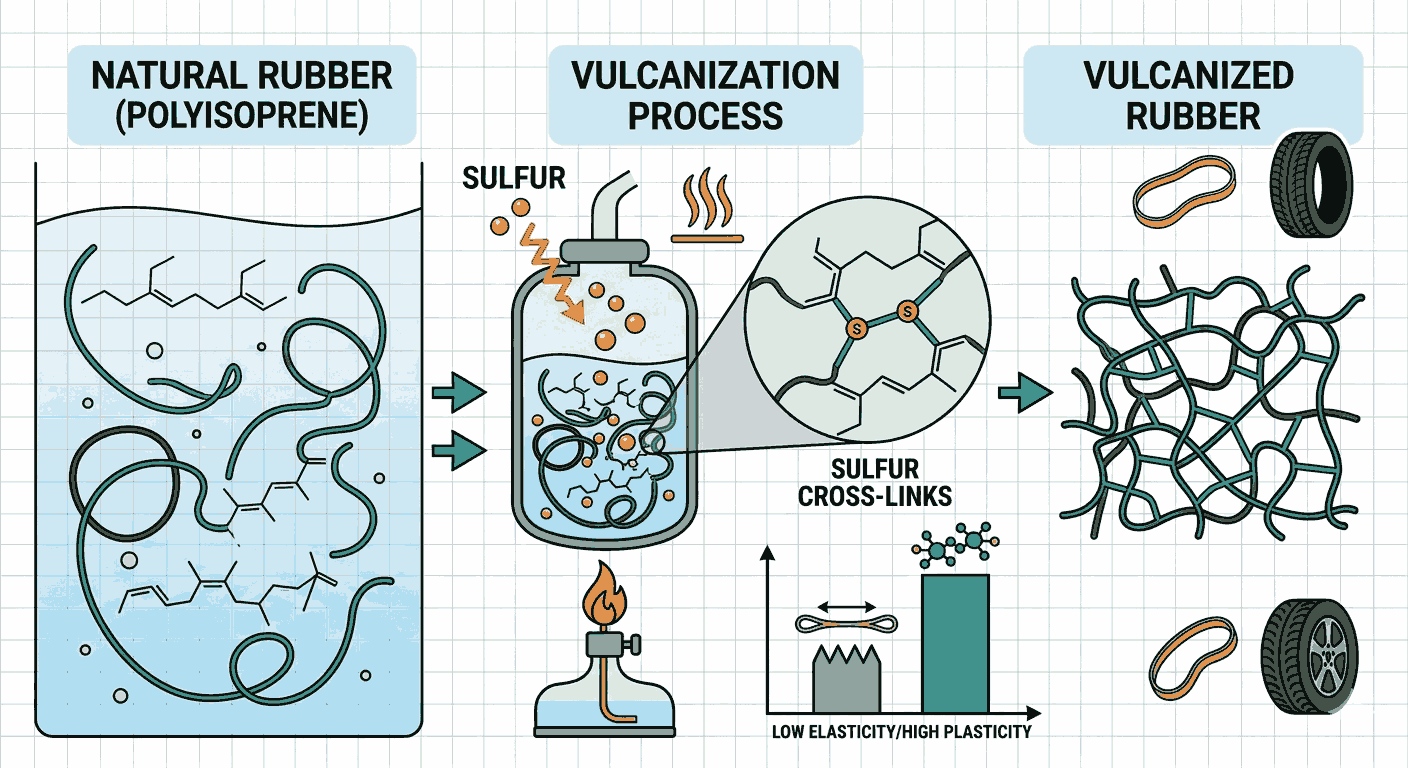
To begin with, Vulcanization and rubber chemistry MCQs with answers help aspirants grasp the structure of natural rubber. Natural rubber is a polymer of isoprene, specifically cis-1,4-polyisoprene. This structure gives rubber its elasticity, but also makes it sensitive to temperature changes, which is why vulcanization becomes important.
When practicing Vulcanization and rubber chemistry MCQs with answers, aspirants learn that vulcanization is a process in which rubber is heated with sulfur. This process introduces cross-links between polymer chains, improving strength, elasticity, and resistance to heat and chemicals.
One of the key reasons to focus on Vulcanization and rubber chemistry MCQs with answers is to understand how the properties of rubber change after vulcanization. Raw rubber is sticky and soft at high temperatures and brittle at low temperatures, whereas vulcanized rubber becomes durable and stable.
Another important concept covered in Vulcanization and rubber chemistry MCQs with answers is the role of sulfur in cross-linking. Sulfur atoms form bridges between polymer chains, restricting their movement and providing improved mechanical properties.
In addition, Vulcanization and rubber chemistry MCQs with answers often emphasize the difference between natural rubber and synthetic rubber. Synthetic rubbers like Buna-S, Buna-N, and neoprene are designed to overcome the limitations of natural rubber and are widely used in industrial applications.
While studying Vulcanization and rubber chemistry MCQs with answers, aspirants also come across elastomers. These are polymers that exhibit elasticity and can return to their original shape after deformation, making them highly useful in products like tires and belts.
Another key area in Vulcanization and rubber chemistry MCQs with answers is the classification of rubber types. Rubber can be classified as natural rubber, synthetic rubber, and reclaimed rubber, each having specific applications based on their properties.
The concept of elasticity is frequently tested in Vulcanization and rubber chemistry MCQs with answers. Elasticity arises due to the coiled structure of polymer chains, which straighten under stress and return to their original form when the stress is removed.
Furthermore, Vulcanization and rubber chemistry MCQs with answers help aspirants understand the industrial importance of vulcanized rubber. It is used in manufacturing tires, footwear, conveyor belts, and many other products due to its enhanced durability.
In competitive exams, Vulcanization and rubber chemistry MCQs with answers often include questions about the chemical reactions involved in rubber formation and modification. Understanding these reactions improves problem-solving ability.
Another significant topic included in Vulcanization and rubber chemistry MCQs with answers is the effect of temperature on rubber properties. Vulcanization reduces sensitivity to temperature variations, making rubber more versatile.
Aspirants practicing Vulcanization and rubber chemistry MCQs with answers also learn about the concept of cross-link density. The number of cross-links determines the hardness and elasticity of the rubber.
Additionally, Vulcanization and rubber chemistry MCQs with answers provide insights into the advantages of synthetic rubber over natural rubber, such as better resistance to oils, chemicals, and environmental conditions.
From an exam perspective, Vulcanization and rubber chemistry MCQs with answers are highly important because they test both theoretical and application-based knowledge.
Regular practice of Vulcanization and rubber chemistry MCQs with answers helps aspirants identify common question patterns and improve their accuracy in exams.
Moreover, Vulcanization and rubber chemistry MCQs with answers support quick revision by summarizing key concepts in a structured manner.
Another benefit of solving Vulcanization and rubber chemistry MCQs with answers is the development of conceptual clarity, especially regarding polymer chemistry and industrial applications.
Aspirants who focus on Vulcanization and rubber chemistry MCQs with answers are better prepared to handle complex questions in exams like NEET, CUET, and JEE.
Furthermore, Vulcanization and rubber chemistry MCQs with answers enhance time management skills during exams, as students become familiar with frequently asked topics.
Vulcanization and Rubber Chemistry MCQs with Answers:
1. Which statement regarding polymers is not correct?
a) Thermosetting polymers are reusable
b) Elastomers have polymer chains held together by weak intermolecular forces
c) Fibers possess high tensile strength
d) Thermoplastic polymers can soften and harden repeatedly
Answer: a
2. Which of the following is NOT a natural polymer?
a) Protein
b) Starch
c) Rubber
d) Rayon
Answer: d
3. Match List I with List II
A → Nylon 6,6
B → Low density polythene
C → High density polythene
D → Teflon
a) III, IV, I, II
b) III, IV, II, I
c) III, I, II, IV
d) II, I, III, IV
Answer: a
4. Melamine polymer is formed by condensation of:
a) Melamine + formaldehyde
b) Phenol + formaldehyde
c) Urea + formaldehyde
d) Styrene + butadiene
Answer: a
5. Which of the following represents neoprene polymer?
a) Polymer of chloroprene
b) Polymer of styrene
c) Polymer of ethylene
d) Polymer of acrylonitrile
Answer: a
6. Which is true about Buna-N?
a) Polymer of butadiene only
b) Copolymer of butadiene and styrene
c) Copolymer of butadiene and acrylonitrile
d) Natural polymer
Answer: c
7.Assertion (A): Dacron is a polyester polymer.
Reason (R): Dacron is formed by condensation of ethylene glycol and terephthalic acid.
a) Both (A) and (R) are correct and (R) is the correct explanation of (A)
b) Both (A) and (R) are correct but (R) is not the correct explanation of (A)
c) (A) is correct but (R) is incorrect
d) (A) is incorrect but (R) is correct
Answer: a
8. The polymer that stretches and regains shape is:
a) Bakelite
b) Nylon 6,6
c) Buna-N
d) Terylene
Answer: c
9. Example of polyester:
a) Buna-S
b) Melamine
c) Neoprene
d) PHBV
Answer: d
10. Conveyor belts are made from:
a) Buna-S
b) Neoprene
c) PVC
d) Styptal
Answer: b
11. Which is NOT a copolymer?
a) Buna-S
b) Neoprene
c) PHBV
d) Butadiene-styrene
Answer: b
12.Assertion (A): Natural rubber is a linear polymer of isoprene (cis-1,4-polyisoprene) showing elasticity.
Reason (R): The polymer chains in natural rubber are coiled and held together by weak intermolecular forces.
a) Both (A) and (R) are true and (R) is the correct explanation of (A)
b) Both (A) and (R) are true but (R) is not the correct explanation of (A)
c) (A) is true but (R) is false
d) (A) is false but (R) is true
Answer: a
13.Which of the following statements regarding polymers is/are correct?
A. Buna-S is a copolymer
B. Nylon-6,6 is a condensation polymer
C. Nylon-6,6 is used as a fibre
D. Terylene is a thermosetting polymer
E. Buna-N is a homopolymer
a) A, B and C
b) B, C and D
c) A, C and E
d) A, B and D
Answer: a
14. Not a condensation polymer:
a) Nylon 6,6
b) Dacron
c) Buna-N
d) Silicon
Answer: c
15. Correctly matched pair:
a) Bakelite – Novolac
b) Polyester – tetrafluoroethene
c) Nylon – acrylonitrile
d) Teflon – caprolactam
Answer: a
16. Novolac polymer mass 963 g, number of monomer units:
a) 9
b) 8
c) 11
d) 7
Answer: a
17. Catalyst for polyethylene manufacture:
a) Ti + Al(CH₃)₃
b) TiCl₄ + C₂H₅MgBr
c) TiCl₃ + Si(CH₃)₃
d) TiCl₄ + Al(CH₃)₃
Answer: d
18.Consider the following statements regarding cellulose:
- Cellulose is a linear biopolymer
- The glucose units are joined by β(1→4) glycosidic linkage
- Cotton and paper are mainly composed of cellulose
Which of the above statements are correct?
a) 1 only
b) 1 and 2
c) 1 and 3
d) 1, 2 and 3
Answer: d
19. Polymer with weakest intermolecular forces:
a) Nylon
b) Terylene
c) Buna-S
d) Polythene
Answer: c
20. Natural rubber is:
a) Polymer of butadiene
b) Polymer of ethylene
c) Polymer of isoprene
d) Polymer of styrene
Answer: c
21. Example of condensation polymer:
a) Nylon
b) Bakelite
c) Urea-formaldehyde
d) All of these
Answer: d
22. Forces present in Nylon 6,6:
a) van der Waals
b) Hydrogen bonding
c) Covalent network
d) None
Answer: b
23. Copolymer example:
a) Polythene
b) Polystyrene
c) Paraformaldehyde
d) Buna-N
Answer: d
24. Polyamide polymer:
a) Terylene
b) Buna-S
c) Polystyrene
d) Nylon 6,6
Answer: d
25. Thermosetting plastics cannot be reshaped due to:
a) van der Waals forces
b) Strong covalent bonds
c) Ionic bonds
d) Hydrogen bonds
Answer: b
26. Nylon-6 is produced by:
a) Addition
b) Copolymerisation
c) Polymerisation
d) Self-condensation
Answer: d
27.Which of the following polymers are biodegradable?
I. PHBV
II. Nylon-2–Nylon-6
III. Glyptal
IV. Bakelite
a) I only
b) II only
c) I and II
d) III and IV
Answer: c
28. Raw material for Nylon-6:
a) Adipic acid
b) Phthalic acid
c) Ethylene glycol
d) Caprolactam
Answer: d
29. Pair of condensation polymers:
a) Terylene and Nylon 6,6
b) PVC and polystyrene
c) Polyvinyl ether and polyisobutene
d) Neoprene and PVP
Answer: a
Conclusion on Vulcanization and Rubber Chemistry MCQs with Answers
In conclusion, Vulcanization and rubber chemistry MCQs with answers are an essential resource for mastering rubber chemistry. They not only improve conceptual understanding but also boost confidence, accuracy, and exam performance.