- Keneitsino Lydia
- March 9, 2026
Ultimate Second Order Reaction MCQs with Answers for Class 12 – Powerful Practice for Competitive Exams
`Chemical kinetics is one of the most important chapters in Class 12 chemistry, and it plays a significant role in understanding how reactions proceed and how reaction rates are determined. Among the various topics, second order reaction MCQs with answers for class 12 are especially useful for strengthening conceptual understanding and problem-solving skills. Practicing second order reaction mcqs with answers class 12 allows aspirants to become familiar with common exam patterns and numerical questions.
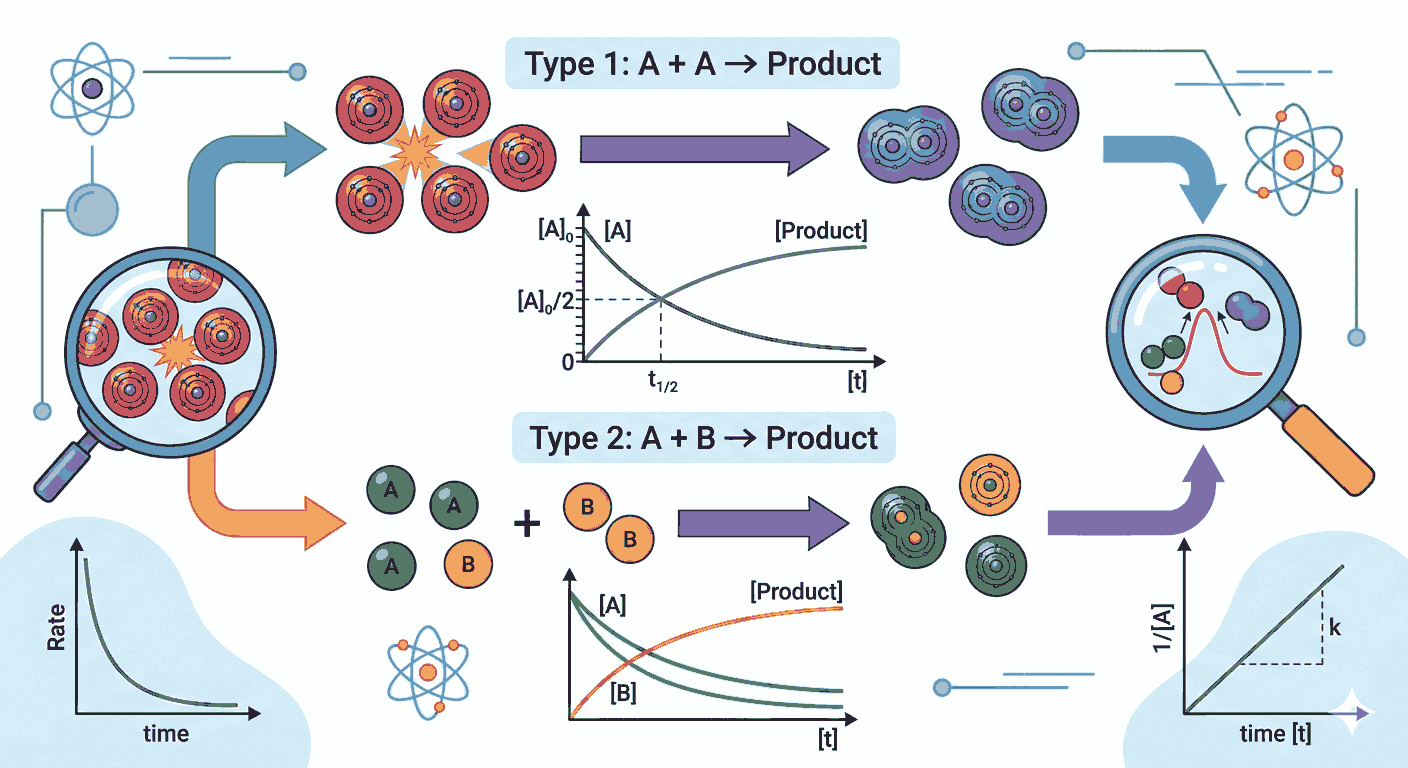
A second order reaction MCQs with answers for class 12 is defined as a reaction whose rate depends on either the square of the concentration of a single reactant or on the product of the concentrations of two reactants. In simple terms, the rate law for such reactions can be expressed as Rate = k[A]² or Rate = k[A][B]. Understanding these relationships becomes easier when aspirants regularly review Second Order Reaction mcqs with answers for class 12, since such practice helps in identifying rate laws and calculating rate constants more efficiently.
One of the distinguishing features of second order reactions is the unit of the rate constant. Unlike first order reactions where the unit is s⁻¹, the unit of the rate constant for second order reactions is generally L mol⁻¹ s⁻¹. This difference is frequently highlighted in Second Order Reaction mcqs with answers for class 12, where aspirants are asked to determine the reaction order from the given unit of the rate constant.
Another important concept related to second order reaction MCQs with answers for class 12 is the integrated rate equation. For a reaction where the rate depends on the square of the concentration of a single reactant, the integrated rate law can be written as
1/[A] = 1/[A]₀ + kt.
This equation helps determine the concentration of the reactant at any time during the reaction. Many Second Order Reaction mcqs with answers for class 12 are based on applying this equation to calculate reaction time or concentration.
Graphical representation is also an important part of studying second order reactions. When the reciprocal of concentration (1/[A]) is plotted against time, a straight line is obtained for a second order reaction. Recognizing this graph is often required when solving Second Order Reaction mcqs with answers for class 12, as it helps identify the reaction order directly from experimental data.
Half-life behavior also differs significantly in second order reactions compared to first order reactions. In second order kinetics, the half-life is inversely proportional to the initial concentration of the reactant. This means that as the initial concentration increases, the half-life decreases. Such relationships are frequently explored in Second Order Reaction mcqs with answers for class 12, helping aspirants understand how concentration influences reaction time.
Temperature also affects second order reactions in the same way it affects other reaction orders. As temperature increases, the rate constant increases according to the Arrhenius equation. In many Second Order Reaction mcqs with answers for class 12, aspirants analyze how temperature changes influence reaction rates and rate constants.
Another key aspect discussed in Second Order Reaction mcqs with answers for class 12 is the difference between molecularity and reaction order. While molecularity refers to the number of reacting molecules in an elementary step, reaction order is determined experimentally from the rate law. A reaction may appear second order based on experimental rate laws even if the overall mechanism involves multiple steps.
Practical examples of second order reaction MCQs with answers for class 12 can be observed in many chemical processes, including bimolecular reactions where two molecules collide to form products. These reactions illustrate how concentration directly influences the reaction rate. Studying such examples through Second Order Reaction mcqs with answers for class 12 allows aspirants to connect theoretical concepts with real chemical behavior.
Second Order Reaction MCQs with Answers for Class 12:
1. Velocity Constant Expression
Q1. The expression for velocity constant for the second order reaction is:
A. k=2.303tlog10aa−xk = \frac{2.303}{t}\log_{10}\frac{a}{a-x}
B. k=1txa[a−x]k = \frac{1}{t}\frac{x}{a[a-x]}
C. k=1tx2a2[a−x]2k = \frac{1}{t}\frac{x^2}{a^2[a-x]^2}
D. None of the above
Answer: B
2. Half-Life of First Order Reaction
Q2. The half-life period for a first order reaction is:
A. Proportional to concentration
B. Independent of concentration
C. Inversely proportional to concentration
D. Inversely proportional to square of concentration
Answer: B
3. Higher Order Reactions
Q3. Higher order (>3) reactions are rare due to:
A. Low probability of simultaneous collision of all reacting species
B. Increase in entropy and activation energy
C. Shifting of equilibrium toward reactants
D. Loss of active species on collision
Answer: A
4. Adsorption of H₂ on Tungsten
Q4. H₂ gas adsorbed on tungsten surface follows:
A. First order
B. Second order
C. Zero order
D. Fifth order
Answer: C
5. Mechanism Question
Q5. For mechanism:
NO + Br₂ ⇌ NOBr₂
NOBr₂ + NO → 2NOBr
If second step is rate determining, order with respect to NO is:
A. 1
B. 2
C. Zero
D. 3
Answer: B
6. Quarter Life of First Order Reaction
Q6. If rate constant = k, the quarter life t1/4t_{1/4} is:
A. 0.75/k
B. 0.69/k
C. 0.29/k
D. 0.10/k
Answer: C
7. Reaction Order with Respect to NO
Q7. For reaction 2NO + 2H₂ → N₂ + 2H₂O, the order with respect to NO is:
A. 1
B. 2
C. 3
D. 4
Answer: A
8. Units of Rate Constants
Q8. For reactions
A → Product (zero order)
B → Product (first order)
Units of k₁ and k₂ respectively are:
A. s⁻¹ , M⁻¹s⁻¹
B. M⁻¹s⁻¹ , M⁻¹s⁻¹
C. None of these
D. Ms⁻¹ , s⁻¹
Answer: D
9. Order from Graph
Q9. The order expected from the given concentration–time relation is:
A. 0
B. 2
C. ∞
D. 1
Answer: D
10. Straight Line [R] vs Time
Q10. If [R] vs time graph is straight with negative slope, reaction order is:
A. Second
B. Third
C. First
D. Zero
Answer: D
11. Time for 75% Completion
Q11. For first order reaction with rate constant 6.909 min⁻¹, time required for 75% completion is:
A. (2/3) log 2
B. (2/3) log 4
C. (3/2) log 2
D. (3/2) log 4
Answer: A
12. Inversion of Cane Sugar
Q12. Inversion of cane sugar in dilute acid is:
A. Bimolecular reaction
B. Pseudo unimolecular reaction
C. Unimolecular reaction
D. Trimolecular reaction
Answer: B
13. Concentration Reduction to 1/8
Q13. For first order reaction with half-life 14 s, time for concentration to become 1/8 is:
A. (14)³ s
B. 28 s
C. 42 s
D. (14)² s
Answer: C
14. Rate Law Problem
Q14. For rate = k[A]ˣ[B]ʸ[C]ᶻ where doubling A, B, C increases rate by 2, 0, 4 respectively, overall order is:
A. 1
B. 2
C. 3
D. 4
Answer: C
15. Rate Constant Units
Q15. If k = 3 × 10⁻³ mol L⁻¹ min⁻¹, order is:
A. Zero
B. 1
C. 2
D. 3
Answer: A
16. Half-Life Independent of Pressure
Q16. If half-life independent of initial pressure, order is:
A. Zero
B. 1
C. 2
D. 3
Answer: B
17. Rate Calculation
Q17. Rate when concentration of X = 0.01 M is:
A. 1.73 × 10⁻⁴ M min⁻¹
B. 3.47 × 10⁻⁴ M min⁻¹
C. 3.47 × 10⁻⁵ M min⁻¹
D. 7.53 × 10⁻⁴ M min⁻¹
Answer: D
18. Temperature Coefficient
Q18. Rate constant at 17°C is:
A. 10²
B. 5 × 10²
C. 2 × 10³
D. 5 × 10⁻⁴
Answer: D
19. Order of Reaction
Q19. Rate = kCA3/2CB−1/2kC_A^{3/2}C_B^{-1/2} order is:
A. 4
B. 1
C. 3
D. 2
Answer: B
20. Order Determination
Q20. Order of reaction is decided by:
A. Temperature
B. Mechanism of reaction
C. Molecularity
D. Pressure
Answer: B
21. Thermal Decomposition
Q21. Time for 90% decomposition if 50% occurs in 120 min is:
A. 300 min
B. 360 min
C. 398.8 min
D. 400 min
Answer: C
22. Zero Order Half-Life
Q22. For zero order reaction:
A. t1/2∝R0t_{1/2} ∝ R_0
B. t1/2∝1/R0t_{1/2} ∝ 1/R_0
C. t1/2∝R02t_{1/2} ∝ R_0^2
D. t1/2∝1/R02t_{1/2} ∝ 1/R_0^2
Answer: A
23. Order of Reaction
Q23. If rate = k[A]¹/²[B]², order is:
A. 0.5
B. 2.5
C. 2
D. 1
Answer: B
24. Half-Life Independent of Concentration
Q24. If half-life independent of concentration, order is:
A. Zero
B. One
C. Two
D. Three
Answer: B
25. Reaction Completion
Q25. If 50% reaction in 25 min, amount reacted in 100 min is:
A. 93.75%
B. 87.5%
C. 75%
D. 100%
Answer: A
26. Partial Pressure of A
Q26. Partial pressure of A is:
A. 90
B. 80
C. 180
D. 100
Answer: B
27. 84% Completion
Q27. Time for 84% completion:
A. 26 min
B. 30 min
C. 40 min
D. 54 min
Answer: C
28. Order Determination
Q28. Reaction order is:
A. Zero
B. 1
C. 2
D. 3
Answer: D
29. Decomposition of AB
Q29. Order for decomposition of AB is:
A. 2
B. 1.5
C. 1
D. Zero
Answer: A
30. Second Order Reaction
Q30. Which of the following is second order?
A. H₂ + Br₂ → 2HBr
B. NH₄NO₃ → N₂ + 3H₂O
C. H₂ + Cl₂ → 2HCl
D. CH₃COOH + NaOH → CH₃COONa + H₂O
Answer: D
Conclusion on Second Order Reaction MCQs with Answers for Class 12
In conclusion, mastering the principles of second order reaction with answers for class 12 is essential for performing well in chemical kinetics. Regular practice using Second Order Reaction mcqs with answers for class 12 helps aspirants develop confidence in solving numerical problems, interpreting graphs, and understanding rate laws. By thoroughly reviewing Second Order Reaction mcqs with answers for class 12, aspirants can strengthen their conceptual clarity and perform better in both board examinations and competitive entrance tests.
