- Keneitsino Lydia
- March 18, 2026
Peptization and Coagulation MCQs Class 12 Chemistry Notes: Master These Powerful Questions for Exam Success
Peptization and coagulation are two fundamental processes in the study of colloids, forming an essential part of Class 12 surface chemistry. These concepts help aspirants understand how colloidal systems are formed, stabilized, and sometimes destabilized. A strong grasp of these ideas is crucial for exams, and many learners reinforce their preparation through Peptization and Coagulation MCQs Class 12 Chemistry Notes. Regular revision using Peptization and Coagulation MCQs Class 12 Chemistry Notes allows aspirants to clearly differentiate between these two opposite processes.
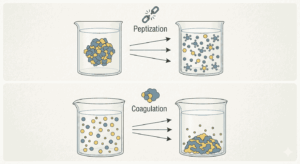
Peptization is the process by which a precipitate is converted into a colloidal solution by the addition of a suitable electrolyte, called a peptizing agent. The electrolyte provides ions that are adsorbed on the surface of the precipitate particles, giving them a charge and causing them to repel each other. This leads to the formation of stable colloidal particles. Understanding this mechanism is essential and is frequently tested through Peptization and Coagulation MCQs Class 12 Chemistry Notes. By practicing Peptization and Coagulation MCQs Class 12 Chemistry Notes, aspirants can easily remember how peptization helps in forming colloidal sols.
On the other hand, coagulation is the process of destabilizing a colloidal solution, resulting in the aggregation and precipitation of colloidal particles. This occurs when the charge on colloidal particles is neutralized, often by the addition of electrolytes. The ions from the electrolyte neutralize the charge, allowing particles to come together and form larger aggregates. This concept is widely covered in Peptization and Coagulation MCQs Class 12 Chemistry Notes, making it a key topic for revision. Practicing Peptization and Coagulation MCQs Class 12 Chemistry Notes helps aspirants understand how coagulation differs from peptization.
The Hardy–Schulze rule plays an important role in coagulation. It states that the effectiveness of an electrolyte in causing coagulation depends on the valency of the ion that has a charge opposite to that of the colloidal particles. Higher the valency, greater is the coagulating power. This rule is commonly tested in exams, and aspirants often revise it using Peptization and Coagulation MCQs Class 12 Chemistry Notes. Repeated exposure to Peptization and Coagulation MCQs Class 12 Chemistry Notes ensures better retention of such rules and their applications.
Another important aspect is the difference in stability between lyophilic and lyophobic sols. Lyophilic sols are more stable due to strong interaction between the dispersed phase and the dispersion medium, while lyophobic sols are less stable and more easily coagulated. This distinction is often highlighted in Peptization and Coagulation MCQs Class 12 Chemistry Notes, helping aspirants avoid confusion. By practicing Peptization and Coagulation MCQs Class 12 Chemistry Notes, aspirants can confidently identify which type of sol is more stable under given conditions.
Coagulation can be achieved through several methods, such as adding electrolytes, electrophoresis, and mixing oppositely charged sols. Each method works by reducing the repulsive forces between particles, leading to aggregation. These methods are frequently included in Peptization and Coagulation MCQs Class 12 Chemistry Notes, making them important for exam preparation. Solving Peptization and Coagulation MCQs Class 12 Chemistry Notes helps aspirants understand which method is applicable in different situations.
Peptization, in contrast, is a method used to prepare colloidal solutions, especially lyophobic sols. It involves converting freshly prepared precipitates into colloidal form by adding small amounts of electrolyte. This process is particularly useful in laboratory preparation of colloids. Questions related to this are commonly found in Peptization and Coagulation MCQs Class 12 Chemistry Notes, emphasizing its importance. Practicing Peptization and Coagulation MCQs Class 12 Chemistry Notes enables aspirants to clearly understand the role of peptizing agents.
In practical applications, coagulation is used in water purification, where impurities are removed by adding coagulants like alum. Peptization is used in industries where stable colloidal solutions are required. These real-life applications are often discussed in Peptization and Coagulation MCQs Class 12 Chemistry Notes, helping aspirants connect theory with practice. Reviewing Peptization and Coagulation MCQs Class 12 Chemistry Notes strengthens both conceptual and practical knowledge.
Peptization and Coagulation MCQs Class 12 Chemistry Notes:
Q1.
Assertion: Fe³⁺ can be used for coagulation of As₂S₃ sol.
Reason: Fe³⁺ reacts with As₂S₃ to give Fe₂S₃.
A. Both Assertion and Reason are correct and Reason is the correct explanation
B. Both Assertion and Reason are correct but Reason is not the correct explanation
C. Assertion is correct but Reason is incorrect
D. Both Assertion and Reason are incorrect
Answer: C
Q2.
Assertion: Colloidal solutions are stable and particles do not settle down.
Reason: Brownian movement counters gravitational force.
A. Both correct and Reason explains Assertion
B. Both correct but Reason not explanation
C. Assertion correct, Reason incorrect
D. Both incorrect
Answer: A
Q3.
Preparation of lyophobic sols by chemical methods involves:
A. Double decomposition
B. Oxidation and reduction
C. Hydrolysis
D. All of these
Answer: D
Q4.
Coagulating values order:
A. NaCl > Na₂SO₄ > Na₃PO₄
B. Na₂SO₄ > Na₃PO₄ > NaCl
C. Na₃PO₄ > Na₂SO₄ > NaCl
D. Na₂SO₄ > NaCl > Na₃PO₄
Answer: A
Q5.
Minimum electrolyte needed to precipitate clay sol:
A. Aluminum sulphate
B. Potassium sulphate
C. Sodium hydroxide
D. Hydrochloric acid
Answer: A
Q6.
Example of associated colloid:
A. Sulphur sol
B. Cellulose
C. Sodium stearate
D. Polystyrene
Answer: C
Q7.
Charge on Fe(OH)₃ sol is due to adsorption of:
A. Cl⁻
B. Fe³⁺
C. OH⁻
D. None
Answer: B
Q8.
Positive AgI sol is formed due to adsorption of:
A. O₃⁻
B. NO₃⁻
C. Ag⁺
D. K⁺
Answer: C
Q9.
An emulsion is:
A. Liquid in gas
B. Liquid in liquid
C. Solid in liquid
D. Gas in solid
Answer: B
Q10.
Property independent of charge:
A. Electrophoresis
B. Electro-osmosis
C. Tyndall effect
D. Coagulation
Answer: C
Q11.
Isoelectric point is when:
A. Particles migrate
B. Particles do not migrate
C. pH = 7
D. pH = 0
Answer: B
Q12.
Not a preparation method:
A. Electrical dispersion
B. Peptization
C. Coagulation
D. Mechanical dispersion
Answer: C
Q13.
Lowest coagulation value:
A. NaCl
B. KCl
C. BaCl₂
D. AlCl₃
Answer: D
Q14.
Incorrect statement:
A. Both sols coagulate by electrophoresis
B. Mixing sols has no effect
C. Na₂SO₄ coagulates both
D. MgCl₂ coagulates gold sol faster
Answer: B
Q15.
Gas dispersed in solid is:
A. Solid sol
B. Gel
C. Aerosol
D. Foam
Answer: A
Q16.
Conditions for Tyndall effect:
A. (A) and (E)
B. (C) and (D)
C. (B) and (D)
D. (B) and (E)
Answer: A
Q17.
Fog is:
A. Gas in liquid
B. Liquid in gas
C. Solid in gas
D. Gas in solid
Answer: B
Q18.
Purification of colloids:
A. Sedimentation
B. Ultrafiltration
C. Filtration
D. Precipitation
Answer: B
Q19.
Best coagulant for Fe(OH)₃:
A. Na₃PO₄
B. NaNO₃
C. Na₂SO₄
D. Na₂HPO₃
Answer: A
Q20.
Helmholtz double layer:
A. AgI : K⁺ : NO₃⁻
B. AgI : Ag⁺ : I⁻
C. AgI : Ag⁺ : NO₃⁻
D. AgI : I⁻ : K⁺
Answer: D
Q21.
Light beam phenomenon:
A. Electrophoresis
B. Electro-osmosis
C. Coagulation
D. Tyndall effect
Answer: D
Q22.
Solid in liquid colloid is:
A. Aerosol
B. Sol
C. Gel
D. Foam
Answer: B
Q23.
Protective action measured by:
A. Oxidation number
B. Atomic number
C. Avogadro number
D. Gold number
Answer: D
Q24.
Not a preparation method:
A. Electrical dispersion
B. Peptization
C. Coagulation
D. Mechanical dispersion
Answer: C
Q25.
Order of protective power:
A. C > B > A
B. A = B = C
C. A > B > C
D. B > A > C
Answer: C
Conclusion on Peptization and Coagulation MCQs Class 12 Chemistry Notes:
Peptization and coagulation are two fundamental processes in the study of colloids, and mastering them is essential for Class 12 Chemistry students. Through peptization and coagulation MCQs class 12 Chemistry notes, aspirants gain a clear understanding of how colloidal particles behave under different conditions. These MCQs not only test theoretical knowledge but also strengthen conceptual clarity, especially regarding the stability and destabilization of colloidal systems.
In peptization and coagulation MCQs class 12 Chemistry notes, peptization is explained as the process of converting a precipitate into a colloidal solution by adding a suitable electrolyte. This highlights the role of common ions in stabilizing dispersed particles. On the other hand, coagulation involves the aggregation and settling of colloidal particles, often caused by the addition of electrolytes or by heating. Understanding these contrasting processes is crucial for solving application-based questions.
By practicing peptization and coagulation MCQs class 12 Chemistry notes, aspirants can easily identify patterns in questions related to Hardy-Schulze rule, charge neutralization, and the role of electrolytes. These MCQs also help in improving speed and accuracy, which is important for competitive exams like NEET and JEE.
Moreover, peptization and coagulation MCQs class 12 Chemistry notes provide a structured approach to revision. They summarize key concepts, highlight frequently asked questions, and ensure that aspirants are well-prepared for both board exams and entrance tests. Regular practice reinforces memory and boosts confidence.
In conclusion, peptization and coagulation MCQs class 12 Chemistry notes serve as an effective tool for mastering colloidal chemistry. They bridge the gap between theory and application, making learning more interactive and exam-oriented. By consistently practicing these MCQs, students can achieve a deeper understanding and perform better in their examinations.
