- Keneitsino Lydia
- April 2, 2026
Master Peptide Formation Fast: 30 Peptide Formation and Amino Acids Chemistry MCQs with Answers for Exam Success
Understanding biomolecules is essential for competitive exams, and Peptide Formation and Amino Acids Chemistry MCQs with Answers play a crucial role in mastering this topic. Amino acids are the building blocks of proteins, and their behavior, reactions, and bonding patterns are frequently tested in exams like NEET, CUET, and JEE.
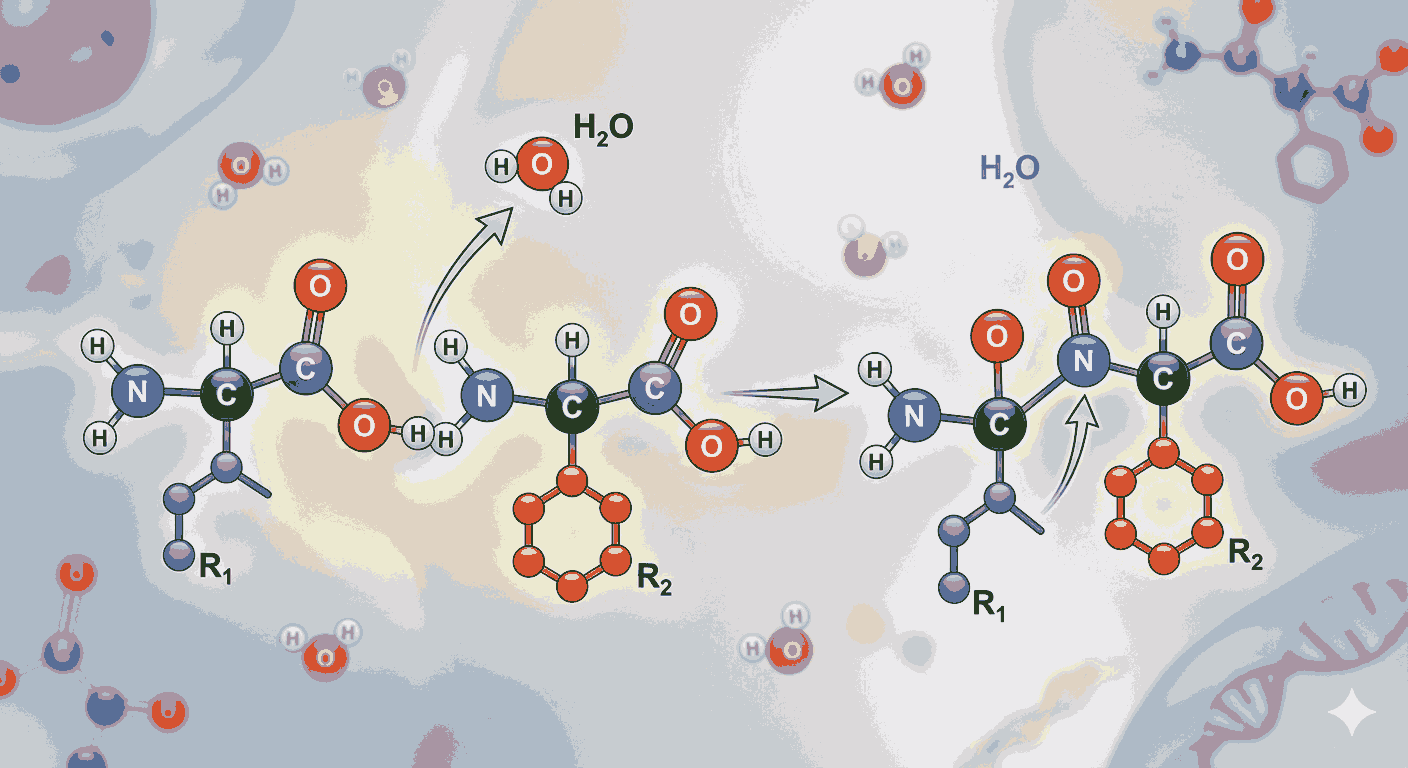
To begin with, Peptide Formation and Amino Acids Chemistry MCQs with Answers help aspirants understand how amino acids combine to form peptides. A peptide bond is formed when the carboxyl group of one amino acid reacts with the amino group of another, releasing a molecule of water. This condensation reaction is fundamental to protein synthesis and is a key concept tested in exams.
When practicing Peptide Formation and Amino Acids Chemistry MCQs with Answers, aspirants often encounter questions related to the structure of amino acids. Each amino acid contains a central carbon atom bonded to an amino group, a carboxyl group, a hydrogen atom, and a variable side chain. This structural variation determines the properties and classification of amino acids.
Another important aspect covered in Peptide Formation and Amino Acids Chemistry MCQs with Answers is the classification of amino acids. They can be categorized as acidic, basic, or neutral depending on the nature of their side chains. This classification directly influences their behavior in different pH environments, which is often tested in conceptual questions.
The concept of zwitter ions is also frequently emphasized in Peptide Formation and Amino Acids Chemistry MCQs with Answers. In aqueous solutions, amino acids exist as dipolar ions, carrying both positive and negative charges. This unique property affects their solubility, melting point, and migration in an electric field.
In addition, Peptide Formation and Amino Acids Chemistry MCQs with Answers often focus on peptide chain formation. When two amino acids combine, they form a dipeptide; three form a tripeptide; and many amino acids form polypeptides. Understanding the number of peptide bonds and their arrangement is a common question type.
Another key topic included in Peptide Formation and Amino Acids Chemistry MCQs with Answers is protein structure. Proteins have primary, secondary, tertiary, and quaternary structures. The primary structure refers to the sequence of amino acids, while higher structures involve folding and interactions like hydrogen bonding and disulfide linkages.
While studying Peptide Formation and Amino Acids Chemistry MCQs with Answers, aspirants also learn about essential and non-essential amino acids. Essential amino acids cannot be synthesized by the human body and must be obtained through diet, making them important from both biological and exam perspectives.
Furthermore, Peptide Formation and Amino Acids Chemistry MCQs with Answers highlight chemical reactions of amino acids, such as reactions with ninhydrin, which produces a characteristic purple color. These reactions are often used in identification-based questions in exams.
Another interesting concept discussed in Peptide Formation and Amino Acids Chemistry MCQs with Answers is the isoelectric point. At this specific pH, an amino acid exists in a neutral zwitterionic form and does not migrate in an electric field. This concept is highly important for understanding amino acid behavior.
Aspirants practicing Peptide Formation and Amino Acids Chemistry MCQs with Answers also become familiar with optical activity. Except for glycine, all amino acids are optically active due to the presence of a chiral carbon atom, which is a frequently tested concept.
Moreover, Peptide Formation and Amino Acids Chemistry MCQs with Answers help in understanding the significance of peptide bonds in biological systems. These bonds are strong and stable, giving proteins their structural integrity.
Another concept reinforced through Peptide Formation and Amino Acids Chemistry MCQs with Answers is hydrolysis of peptides. Under acidic or enzymatic conditions, peptide bonds can break down to yield individual amino acids.
In competitive exams, Peptide Formation and Amino Acids Chemistry MCQs with Answers are valuable for improving problem-solving speed. They help students recognize patterns in questions and apply concepts quickly and accurately.
Practicing Peptide Formation and Amino Acids Chemistry MCQs with Answers regularly also enhances conceptual clarity. Instead of rote learning, aspirants develop a deeper understanding of reaction mechanisms and molecular behavior.
Additionally, Peptide Formation and Amino Acids Chemistry MCQs with Answers are useful for revision. They provide a quick and efficient way to revisit important topics before exams.
From an exam perspective, Peptide Formation and Amino Acids Chemistry MCQs with Answers often include assertion-reason questions, matching-type questions, and application-based problems, making them essential for thorough preparation.
By solving Peptide Formation and Amino Acids Chemistry MCQs with Answers, aspirants can identify their weak areas and focus on improving them systematically.
Furthermore, Peptide Formation and Amino Acids Chemistry MCQs with Answers help in building confidence, especially when dealing with complex biomolecule questions.
Peptide Formation and Amino Acids Chemistry MCQs with Answers:
1. Which of the following is not an essential amino acid?
a) Leucine
b) Valine
c) Lysine
d) Tyrosine
Answer: d
2.Which of the following represents the correct structure of tyrosine?
a) HO–C₆H₄–CH₂–CH(NH₂)–COOH
b) C₆H₅–CH₂–CH(NH₂)–COOH
c) HO–C₆H₄–CH(NH₂)–COOH
d) HO–C₆H₄–CH₂–CH₂–COOH
Answer: a
3. Total negative charge in Gly–Glu–Asp–Tyr at pH 12.5
a) 2
b) 3
c) 4
d) 5
Answer: c
4. Number of peptide linkages
a) 4
b) 3
c) 2
d) 1
Answer: b
5. Number of amino groups
a) 6
b) 2
c) 5
d) 4
Answer: c
6. Essential amino acid
a) Phenylalanine
b) Cysteine
c) Alanine
d) Glycine
Answer: a
7. Product after DNFB reaction
a) R-CH(NH₂)-COOH
b) NH₂CHR²COOH
c) Mixed product
d) NH₂-CR₂’-COOH
Answer: b
8. Nature at pH < pKa
a) Anion
b) Cation
c) Zwitterion
d) Neutral
Answer: b
9. No ninhydrin purple colour
a) Proline
b) Glycine
c) Lysine
d) Aspartic acid
Answer: a
10. Product with benzoyl chloride
a) PhCOCH₂NH₂
b) PhCH₂NH₂
c) PhCONHCH₃
d) PhCONHCH₂COOH
Answer: d
11. Peptides are formed from
a) Amines
b) Carbohydrates
c) α-amino acids
d) Aromatic amines
Answer: c
12. DNA pairing
a) A–T, G–C
b) A–C, G–T
c) A–G, C–T
d) A–U, G–C
Answer: a
13. Chiral amino acid
a) Ethylalanine
b) Methylglycine
c) Hydroxymethyl serine
d) Tryptophan
Answer: c
14. Aromatic amino acid
a) Lysine
b) Serine
c) Tyrosine
d) Glutamine
Answer: c
15. Isoelectric point
a) 4.32
b) 3.34
c) 9.46
d) 5.97
Answer: d
16. Optically inactive amino acid
a) Phenylalanine
b) Glycine
c) Glutamic acid
d) Asparagine
Answer: b
17. Essential amino acid
a) Tyrosine
b) Cysteine
c) Isoleucine
d) Serine
Answer: c
18. Molar mass of protein
a) 36500
b) 35400
c) 34800
d) 35600
Answer: d
19. Glycine in alkaline medium
a) Cation
b) Anion
c) Zwitter ion
d) Covalent
Answer: b
20. Max tripeptides
a) 4000
b) 6470
c) 7465
d) 8000
Answer: d
21. Not optically active
a) Lactic acid
b) Serine
c) Alanine
d) Glycine
Answer: d
22. Chirality in DNA
a) D-sugar
b) L-sugar
c) Bases
d) Phosphate
Answer: a
23. Amino acids in insulin
a) 25
b) 51
c) 20
d) 22
Answer: b
24. Mercapto group amino acid
a) Leucine
b) Glutamine
c) Cysteine
d) Lysine
Answer: c
25. Protein is polymer of
a) Carbohydrates
b) Amino acids
c) Nucleic acids
d) Acids
Answer: b
26. Base only in RNA
a) Guanine
b) Adenine
c) Cytosine
d) Uracil
Answer: d
27. Vasodilator
a) Histamine
b) Serotonin
c) Codeine
d) Cimetidine
Answer: a
28. Not basic amino acid
a) Aspartic acid
b) Lysine
c) Arginine
d) Histidine
Answer: a
29. Building units of protein
a) α-amino acids
b) β-amino acids
c) γ-amino acids
d) None
Answer: a
30. Glycine synthesis
a) Strecker
b) Williamson
c) Perkin
d) Coupling
Answer: a
31. Basic amino acid
a) Histidine
b) Tyrosine
c) Proline
d) Valine
Answer: a
32. Basic amino acid
a) Valine
b) Tyrosine
c) Arginine
d) Leucine
Answer: c
33. Forms zwitter ion
a) Aniline
b) Acetanilide
c) Benzoic acid
d) Glycine
Answer: d
34. Linkage in proteins
a) Peptide bond
b) Dative
c) Glycosidic
d) β-glycosidic
Answer: a
35. Basic amino acid
a) Serine
b) Alanine
c) Tyrosine
d) Lysine
Answer: d
36. Non-essential amino acid
a) Lysine
b) Valine
c) Leucine
d) Alanine
Answer: d
37. Nonessential amino acid
a) Valine
b) Proline
c) Lysine
d) Phenylalanine
Answer: b
38. Product with nitrous acid
a) Propane-2-ol
b) Cyclopropene
c) Propanol
d) Nitropropane
Answer: a
39.Which of the following represents the correct structure of the dipeptide Gly–Ala?
a) NH₂–CH₂–CO–NH–CH(CH₃)–COOH
b) NH₂–CH(CH₃)–CO–NH–CH₂–COOH
c) NH₂–CH₂–COOH–NH–CH(CH₃)–COOH
d) NH₂–CH₂–CO–CH(CH₃)–NH–COOH
Answer: a
40. DNA linkage
a) Phosphodiester
b) Monoester
c) Triester
d) Amide
Answer: a
41.In a linear tetrapeptide made of different amino acids, what are the number of amino acids and peptide bonds respectively?
a) 4 and 4
b) 5 and 5
c) 5 and 4
d) 4 and 3
Answer: d
42. At different pH values, alanine exists in different forms. Which pair correctly represents alanine at pH 2–4 and pH 9–11 respectively?
I. H₃N⁺–CH(CH₃)–COOH
II. H₂N–CH(CH₃)–COO⁻
III. H₃N⁺–CH(CH₃)–COO⁻
IV. H₂N–CH(CH₃)–COOH
a) I and II
b) I and III
c) II and III
d) III and IV
Answer: a
Conclusion on Peptide Formation and Amino Acids Chemistry MCQs with Answers
In conclusion, Peptide Formation and Amino Acids Chemistry MCQs with Answers are an indispensable resource for aspirants preparing for competitive exams. They not only strengthen conceptual understanding but also improve accuracy and speed, ensuring better performance in exams.
