- Keneitsino Lydia
- March 11, 2026
Powerful Practice Set – 30 Minimum Energy for Reaction MCQs with Answers for NEET, JEE & CUET Aspirants
In Minimum energy for reaction mcqs with answers ,Chemical kinetics is an essential part of chemistry that explains how quickly reactions occur and what factors influence their speed. One of the most important ideas in kinetics is the concept of minimum energy for reaction mcqs with answers required for a reaction to occur. In many chemistry exams and competitive tests, aspirants frequently practice Minimum Energy for Reaction MCQs with Answers to understand this concept more clearly. Studying Minimum Energy for Reaction MCQs with Answers helps aspirants connect theoretical knowledge with practical problem-solving in reaction kinetics.
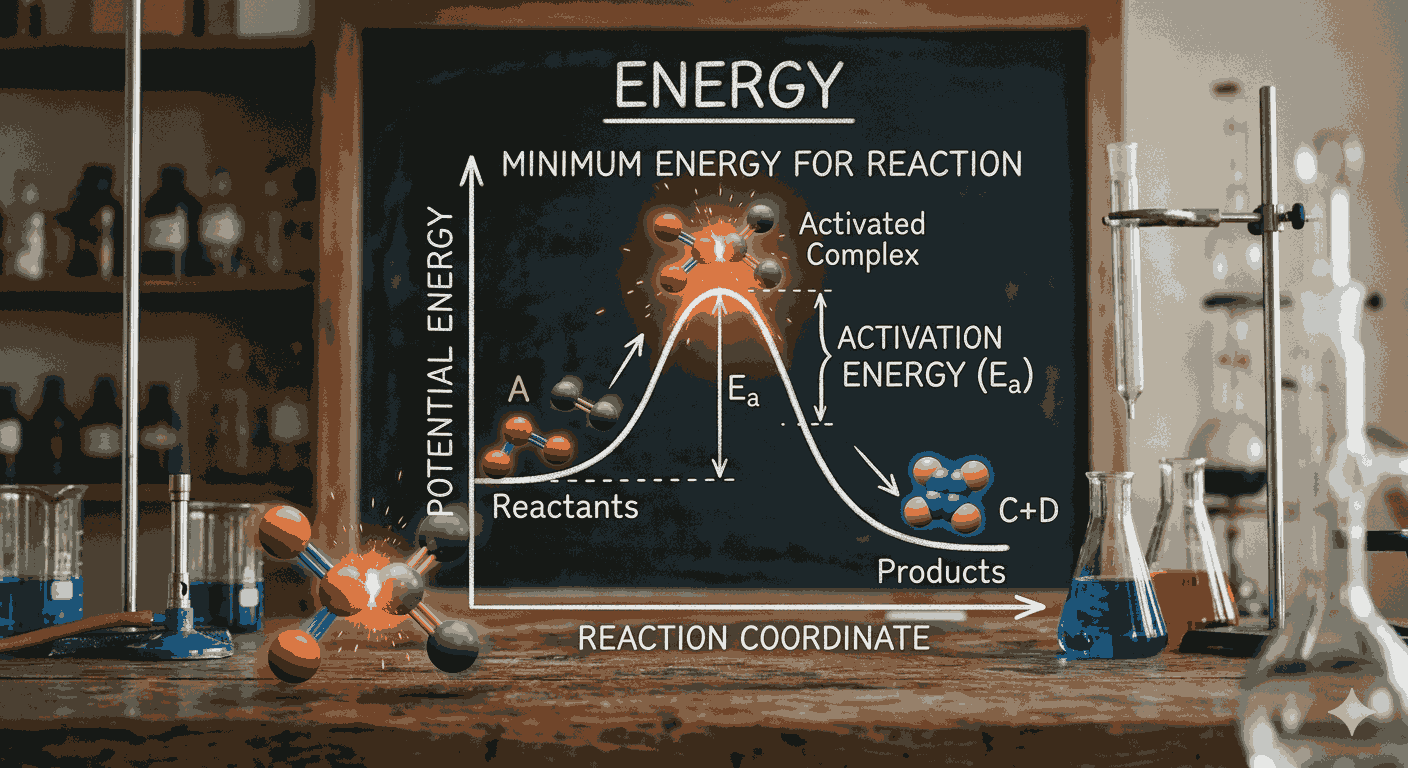
The minimum energy for reaction mcqs with answers required for a chemical reaction to take place is called activation energy. Reactant molecules must possess this minimum amount of energy in order to form an activated complex and convert into products. Without reaching this energy threshold, even frequent molecular collisions will not lead to a successful reaction. This fundamental idea is commonly tested in Minimum Energy for Reaction MCQs with Answers, where aspirants are asked to identify how activation energy influences reaction rate.
According to collision theory, molecules must collide with sufficient energy and proper orientation to react. When temperature increases, the number of molecules possessing the minimum energy required for reaction also increases. This leads to an increase in the reaction rate. Such relationships between temperature and activation energy are frequently explored through Minimum Energy for Reaction MCQs with Answers, allowing aspirants to understand why reactions proceed faster at higher temperatures.
The mathematical relationship between reaction rate and activation energy is explained through the Arrhenius equation. This equation shows how the rate constant depends on temperature and activation energy.
k = Ae^{-\frac{E_a}{RT}}
In this expression, kk is the rate constant, AA is the frequency factor, EaE_a represents activation energy, RR is the gas constant, and TT is the temperature in Kelvin. When aspirants solve Minimum Energy for Reaction MCQs with Answers, they often apply this equation to determine how temperature changes influence reaction speed. Practicing Minimum Energy for Reaction MCQs with Answers also helps aspirants understand how small variations in temperature can significantly increase reaction rates.
Graphical interpretation is another important tool used in chemical kinetics. When the natural logarithm of the rate constant is plotted against the inverse of temperature, a straight line is obtained. The slope of this line is related to activation energy. This graphical method is widely used in laboratory experiments and is also a common topic in Minimum Energy for Reaction MCQs with Answers. By interpreting such graphs, aspirants can determine activation energy and better understand the temperature dependence of reaction rates.
Another factor closely related to activation energy is the role of catalysts. Catalysts increase the rate of a reaction by providing an alternative pathway with lower activation energy. They do not change the overall energy difference between reactants and products, but they make it easier for molecules to reach the activated state. Many Minimum Energy for Reaction MCQs with Answers focus on comparing catalyzed and uncatalyzed reactions to show how lowering activation energy accelerates chemical reactions.
Activation energy also explains why some reactions occur very slowly under normal conditions. Even if molecules collide frequently, the reaction will not proceed unless they possess sufficient energy to overcome the activation barrier. This concept is particularly important in industrial chemistry, where controlling activation energy can improve reaction efficiency. Understanding these principles through Minimum Energy for Reaction MCQs with Answers allows aspirants to appreciate the practical applications of chemical kinetics.
Biological systems also rely heavily on activation energy concepts. Enzymes act as biological catalysts that lower activation energy, enabling biochemical reactions to occur rapidly at normal body temperatures. Without enzymes, many essential biochemical reactions would occur far too slowly to sustain life. This biological relevance is often discussed in Minimum Energy for Reaction MCQs with Answers, highlighting the interdisciplinary importance of activation energy in chemistry and biology.
Practicing Minimum energy for reaction mcqs with answers allows learners to connect theoretical concepts with real biological processes.In many competitive exams, minimum energy for reaction mcqs with answers are used to test the understanding of enzyme activity and reaction rates in biological systems. Therefore, solving minimum energy for reaction mcqs with answers can strengthen both chemistry and biology concepts together.
Another key concept associated with activation energy is the energy profile diagram of a reaction. These diagrams show the energy changes that occur during a reaction, including the energy of reactants, the activated complex, and the products. The peak of the energy curve represents the activation energy barrier. Understanding these diagrams is essential for solving conceptual problems found in Minimum Energy for Reaction MCQs with Answers, as they visually illustrate how energy influences reaction progress.
Minimum Energy for Reaction MCQs with Answers:
1. Excess energy required for reaction
Q1. The excess energy which a molecule must possess to become active is known as:
A. Kinetic energy
B. Threshold energy
C. Potential energy
D. Activation energy
Answer: D
2. Apparent rate constant
Q2. For the conversion of chair form to boat form, e−Ea/RT=4.35×10−8e^{-E_a/RT} = 4.35 \times 10^{-8} at 298 K with a pre-exponential factor 1012 s−110^{12}\ \text{s}^{-1}. The apparent rate constant at 298 K is:
A. 4.35×104 s−14.35 \times 10^{4}\ \text{s}^{-1}
B. 4.2×105 s−14.2 \times 10^{5}\ \text{s}^{-1}
C. 4.2×106 s−14.2 \times 10^{6}\ \text{s}^{-1}
D. 4.35×103 s−14.35 \times 10^{3}\ \text{s}^{-1}
Answer: B
3. Activation energy in exothermic reaction
Q3. For an exothermic reaction A⇌BA \rightleftharpoons B, if EfE_f and EbE_b are the activation energies of forward and backward reactions respectively, which is correct?
A. Ef>EbE_f > E_b
B. Ef=EbE_f = E_b
C. Ef<EbE_f < E_b
D. Ef=−EbE_f = -E_b
Answer: C
4. Reverse activation energy
Q4. An endothermic reaction A→BA \to B has activation energy x kJ/molx\ \text{kJ/mol}. If the energy change is y kJy\ \text{kJ}, the activation energy of the reverse reaction is:
A. x+yx + y
B. x−yx – y
C. y−xy – x
D. −x-x
Answer: B
5. Assertion–Reason
Q5. Assertion: According to transition state theory, one vibrational degree of freedom converts to translational degree.
Reason: Energy of activated complex is higher than reactants.
A. Both correct, Reason explains Assertion
B. Both correct, Reason does not explain Assertion
C. Assertion correct, Reason wrong
D. Both wrong
Answer: B
6. Comparison of activation energies
Q6. If rate constant k1k_1 is double k2k_2, the activation energies Ea1E_{a1} and Ea2E_{a2} satisfy:
A. Ea1<Ea2E_{a1} < E_{a2}
B. Ea1>Ea2E_{a1} > E_{a2}
C. Ea1=Ea2E_{a1} = E_{a2}
D. Ea1=2Ea2E_{a1} = 2E_{a2}
Answer: A
7. Ratio of rate constant to Arrhenius factor
Q7. If activation energy =2.303RT= 2.303RT, the ratio of rate constant to Arrhenius factor is:
A. 10−110^{-1}
B. 10−210^{-2}
C. 10
D. 2×10−32 \times 10^{-3}
Answer: A
8. Parallel reactions
Q8. If Ea2=2Ea1E_{a2} = 2E_{a1}, the relation between k1k_1 and k2k_2 is:
A. k1=2k2eEa2/RTk_1 = 2k_2e^{E_{a2}/RT}
B. k1=k2eEa1/RTk_1 = k_2e^{E_{a1}/RT}
C. k2=k1eEa2/RTk_2 = k_1e^{E_{a2}/RT}
D. k1=Ak2eEa1/RTk_1 = Ak_2e^{E_{a1}/RT}
Answer: B
9. Activation energy from temperature rise
Q9. If rate doubles when temperature increases from 300 K to 310 K, activation energy is:
A. 53.6 kJ mol⁻¹
B. 48.6 kJ mol⁻¹
C. 58.5 kJ mol⁻¹
D. 65 kJ mol⁻¹
Answer: A
10. Negative activation energy reactions
Q10. Reactions with negative activation energy:
A. Decrease with increasing temperature
B. Increase with increasing temperature
C. Independent of temperature
D. Depend on potential barrier height
Answer: A
11. Reverse activation energy for exothermic reaction
Q11. For reaction A+B→C+D+40 kJA + B \rightarrow C + D + 40\ \text{kJ} with activation energy 18 kJ, the reverse activation energy is:
A. 58 kJ
B. 40 kJ
C. 18 kJ
D. 22 kJ
Answer: A
12. Backward activation energy formula
Q12. If forward activation energy = yy and enthalpy change = −x-x, backward activation energy is:
A. y−xy – x
B. y+xy + x
C. x−yx – y
D. 2x2x
Answer: A
13. Determining activation energy
Q13. Activation energy of a reaction can be determined by:
A. Rate constant at standard conditions
B. Rate constants at two temperatures
C. Rate constant of forward reaction
D. Using catalyst
Answer: B
14. Half-life calculation
Q14. The half-life of a first order reaction with Ea=39.3 kcal mol−1E_a = 39.3\ \text{kcal mol}^{-1} is:
A. 6 h
B. 12 h
C. 1.68 h
D. 1.1 h
Answer: C
15. Arrhenius straight-line plot
Q15. A straight line is obtained by plotting:
A. 1/T1/T
B. log T
C. T
D. log(1/T)
Answer: A
16. Activation energy from slope
Q16. If slope of 1/T1/T vs ln k is −104-10^4, activation energy is:
A. 83.14 kJ mol⁻¹
B. 120.2 kJ mol⁻¹
C. 12.02 mol⁻¹
D. 313.4 kJ mol⁻¹
Answer: A
17. Souring of milk
Q17. Activation energy for souring of milk stored longer in refrigerator is:
A. 2.303×102.303 \times 10 kJ mol⁻¹
B. 2.303×2.7R2.303 \times 2.7R kJ mol⁻¹
C. 2.303×10R2.303 \times 10R kJ mol⁻¹
D. 2.303×2.72.303 \times 2.7 kJ mol⁻¹
Answer: B
18. Ratio of rate constant to Arrhenius factor
Q18. The activation energy of a reaction at a given temperature is 2.303RT J mol−12.303RT\ \text{J mol}^{-1}. The ratio of rate constant to Arrhenius factor is:
A. 0.01
B. 0.1
C. 0.001
D. 2
Answer: B
19. Effect of temperature on reactions with different activation energies
Q19. The activation energies of two reactions are E1E_1 and E2E_2 where E1>E2E_1 > E_2. If temperature increases from T1T_1 to T2T_2, which relation is correct?
A. k1′k1=k2′k2\frac{k_1′}{k_1} = \frac{k_2′}{k_2}
B. k1′k1>k2′k2\frac{k_1′}{k_1} > \frac{k_2′}{k_2}
C. k1′k1<k2′k2\frac{k_1′}{k_1} < \frac{k_2′}{k_2}
D. k1′k1=k2′k2=1\frac{k_1′}{k_1} = \frac{k_2′}{k_2} = 1
Answer: B
20. Temperature where rate constants are equal
Q20. For reactions:
A→B, k1=1010e−20000/TA \rightarrow B,\ k_1 = 10^{10}e^{-20000/T}
A→D, k2=1012e−24606/TA \rightarrow D,\ k_2 = 10^{12}e^{-24606/T}
The temperature at which k1=k2k_1 = k_2 is:
A. 400 K
B. 1000 K
C. 800 K
D. 1500 K
Answer: B
21. Temperature where two rate constants are equal
Q21. If
k1=1016e−2000/Tk_1 = 10^{16} e^{-2000/T}
k2=1015e−1000/Tk_2 = 10^{15} e^{-1000/T}
The temperature at which k1=k2k_1 = k_2 is:
A. 2000 K
B. 10002.303K\frac{1000}{2.303} K
C. 2.303 K
D. 20002.303K\frac{2000}{2.303} K
Answer: B
22. Activation energy from temperature change
Q22. What is the activation energy for a reaction if its rate doubles when the temperature is raised from 20°C to 35°C?
(R=8.314 J mol−1K−1)(R = 8.314\ \text{J mol}^{-1}\text{K}^{-1})
A. 34.7 kJ mol⁻¹
B. 15.7 kJ mol⁻¹
C. 34.2 kJ mol⁻¹
D. 269 kJ mol⁻¹
Answer: A
23. Intercept in Arrhenius plot
Q23. In an Arrhenius plot, the intercept is equal to:
A. −Ea/R-E_a/R
B. lnA\ln A
C. lnk\ln k
D. log10a\log_{10} a
Answer: B
24. Minimum energy for reaction
Q24. The minimum energy required for reacting molecules to undergo a chemical reaction is called:
A. Potential energy
B. Kinetic energy
C. Thermal energy
D. Activation energy
Answer: D
25. Ratio of rate constants at two temperatures
Q25. For a chemical reaction at 27°C, the activation energy is 600R. The ratio of rate constants at 327°C and 27°C will be:
A. 2
B. 20
C. e
D. e²
Answer: C
26. Energy diagram comparison
Q26. From the given energy diagrams for reactions M and N, which statement is correct?
A. Reaction M is faster and less exothermic than reaction N
B. Reaction N is slower and less exothermic than reaction M
C. Reaction M is faster and more exothermic than reaction N
D. Reaction M is slower and more exothermic than reaction N
Answer: C
27. Half-life from pressure graph
Q27. For the decomposition of azomethane
CH3N=NCH3(g)→CH3CH3(g)+N2(g)CH_3N=NCH_3(g) \rightarrow CH_3CH_3(g) + N_2(g)
The reaction follows first-order kinetics. The half-life of the reaction is:
A. 5×10−55 \times 10^{-5} s
B. 4×10−54 \times 10^{-5} s
C. 3×10−53 \times 10^{-5} s
D. 2×10−52 \times 10^{-5} s
Answer: D
28. Order of reaction from half-life data
Q28. The half-life for decomposition of gaseous compound A is 240 s when pressure is 500 Torr. When pressure is 250 Torr, the half-life becomes 4 min.
The order of the reaction is:
A. 1
B. 2
C. 3
D. 4
Answer: A
29. Time for 80% decomposition
Q29. At 30°C, the half-life for decomposition of AB₂ is 200 s and is independent of initial concentration.
The time required for 80% decomposition is:
A. 233 s
B. 323 s
C. 467 s
D. 523 s
Answer: C
30. Relation between completion time and half-life
Q30. The time required for 93.75% completion of a first-order reaction is x minutes. The half-life is:
A. x/3
B. x/2
C. x/4
D. x/8
Answer: C
Conclusion on Minimum Energy for Reaction MCQs with Answers
In conclusion, activation energy represents the minimum energy for reaction mcqs with answers required for a chemical reaction to occur. It plays a critical role in determining reaction rates, temperature dependence, and the effectiveness of catalysts. By regularly practicing Minimum Energy for Reaction MCQs with Answers, aspirants can strengthen their understanding of activation energy and chemical kinetics. Consistent practice with Minimum Energy for Reaction MCQs with Answers not only improves conceptual clarity but also prepares aspirants for exam questions related to reaction mechanisms, temperature effects, and catalytic processes.
