- Keneitsino Lydia
- March 26, 2026
Disaccharides Polysaccharides MCQs Advanced Level – Proven Questions for Top Rankers
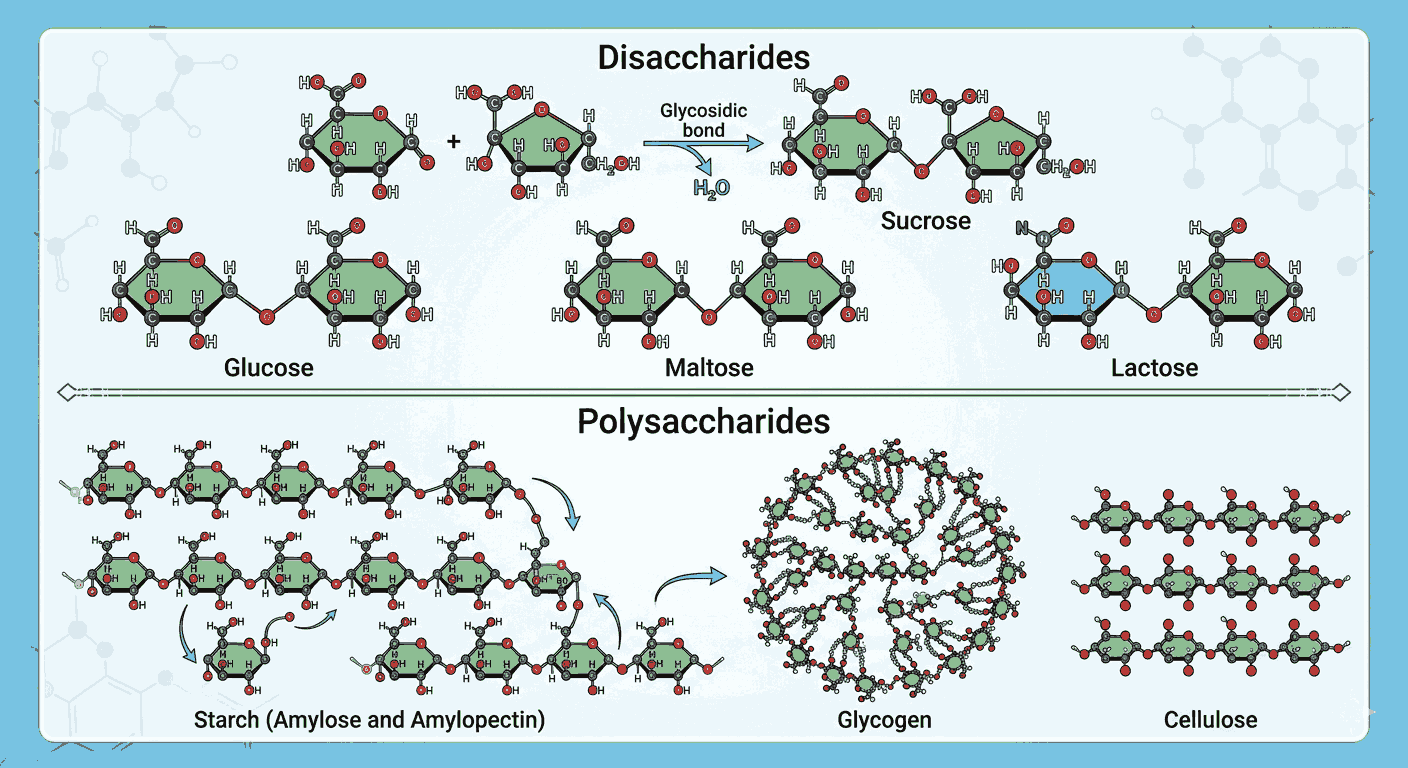
Understanding carbohydrates at a deepenr level is essential for aspirants preparing for competitive exams like NEET, JEE, and CUET. The topic of disaccharides and polysaccharides often requires conceptual clarity and application-based thinking. Practicing Disaccharides polysaccharides mcqs advanced level helps aspirants strengthen their grasp on structures, linkages, and biochemical behavior.
When aspirants engage with Disaccharides polysaccharides mcqs advanced level, they begin to understand how glycosidic linkages define the properties of sugars. Concepts such as α- and β-linkages, reducing and non-reducing sugars, and structural differences between starch, cellulose, and glycogen become clearer through repeated exposure.
One of the main advantages of solving Disaccharides polysaccharides mcqs advanced level is the ability to identify subtle differences between molecules. For instance, differentiating between amylose and amylopectin or lactose and sucrose becomes easier when aspirants actively apply their knowledge. This type of practice also helps in recognizing how branching affects solubility and digestibility.
In addition, Disaccharides polysaccharides mcqs advanced level are particularly useful for mastering enzyme-related reactions. Questions often involve hydrolysis, fermentation, or enzymatic breakdown, which are common in exams. By working through these scenarios, aspirants improve their analytical skills and reaction-based understanding.
Another important aspect covered through Disaccharides polysaccharides mcqs advanced level is stereochemistry. The role of anomeric carbon, mutarotation, and epimerization becomes more intuitive when aspirants repeatedly encounter these ideas in question form. This builds confidence in tackling tricky conceptual problems.
Practicing Disaccharides polysaccharides mcqs advanced level also enhances memory retention. Instead of memorizing facts, aspirants learn patterns and relationships between different carbohydrates. This leads to long-term understanding, which is crucial for higher-level exams.
Moreover, Disaccharides polysaccharides mcqs advanced level allow aspirants to connect theoretical knowledge with real-world biological applications. For example, understanding glycogen as animal starch or cellulose as a structural component of plants becomes more meaningful when linked with practical questions.
Time management is another benefit of solving Disaccharides polysaccharides mcqs advanced level. Aspirants learn to quickly analyze and eliminate incorrect options, a skill that is essential during competitive exams. This repeated practice improves speed and accuracy simultaneously.
In advanced-level preparation, Disaccharides polysaccharides mcqs advanced level often include multi-step reasoning. These questions require aspirants to combine multiple concepts, such as structure, reactivity, and biological function. This holistic approach ensures thorough preparation.
Another key advantage of Disaccharides polysaccharides mcqs advanced level is that they expose aspirants to exam patterns. Many competitive exams reuse similar concepts with slight variations. Practicing these MCQs prepares aspirants to handle unfamiliar questions with confidence.
Additionally, Disaccharides polysaccharides mcqs advanced level help aspirants identify their weak areas. By analyzing mistakes, they can focus on improving specific topics like glycosidic bonds or carbohydrate classification.
Regular practice of Disaccharides polysaccharides mcqs advanced level also boosts problem-solving confidence. As aspirants become familiar with different question formats, they develop a systematic approach to solving complex problems.
Furthermore, Disaccharides polysaccharides mcqs advanced level play a crucial role in revision. They provide a quick and effective way to revisit important concepts before exams. This makes them an essential tool for last-minute preparation.
Aspirants who consistently solve Disaccharides polysaccharides mcqs advanced level tend to perform better because they develop a deeper conceptual understanding rather than relying on rote learning.
In competitive exams, questions from carbohydrates are often tricky. Practicing Disaccharides polysaccharides mcqs advanced level ensures that aspirants are well-prepared for such challenges.
Another benefit of Disaccharides polysaccharides mcqs advanced level is that they encourage logical thinking. Instead of guessing answers, students learn to apply concepts systematically.
By working on Disaccharides polysaccharides mcqs advanced level, aspirants also improve their ability to interpret diagrams and structures, which is an important skill in organic chemistry.
Consistency is key when it comes to mastering carbohydrates. Regular exposure to Disaccharides polysaccharides mcqs advanced level ensures steady progress and better retention.
Additionally, Disaccharides polysaccharides mcqs advanced level help bridge the gap between basic theory and advanced applications, making them ideal for high-level exam preparation.
Aspirants aiming for top ranks should prioritize solving Disaccharides polysaccharides mcqs advanced level as part of their daily study routine.
Disaccharides Polysaccharides MCQs Advanced Level:
1. What percentage of β-D-(+)-glucopyranose is found at equilibrium in aqueous solution?
A. 64 %
B. 36 %
C. ~100 %
D. ~50 %
Answer: A
2. The general molecular formula for disaccharide is:
A. C₁₂H₂₄O₁₁
B. C₆H₁₀O₅
C. C₆H₁₂O₆
D. C₁₂H₂₂O₁₁
Answer: D
3. Which of the following is correct?
A. Catalyst undergoes permanent chemical change
B. Particle size of solute in true solution is 10⁻³ nm
C. Starch solution is a hydrosol
D. Hydrolysis of liquid ester is heterogeneous catalysis
Answer: C
4. Which of the following does not reduce Fehling’s solution?
A. Benzaldehyde
B. Formic acid
C. Glucose
D. Fructose
Answer: D
5. Glucose differs from fructose in that it:
A. Does not undergo hydrolysis
B. Gives silver mirror with Tollen’s reagent
C. Is a monosaccharide
D. None of these
Answer: B
6. Saccharin is manufactured from:
A. Toluene
B. Cyclohexane
C. Starch
D. Cellulose
Answer: A
7. Which of the following act as epimeric pair?
A. Glucose and fructose
B. Fructose and mannose
C. Glucose and mannose
D. Glucose and sucrose
Answer: C
8. Invert sugar is a mixture of:
A. D-glucose + D-fructose
B. D-glucose + D-galactose
C. L-glucose + D-fructose
D. L-glucose + L-fructose
Answer: A
9. Correct structure of cellulose:
A. Branched α (1→4) & α (1→6)
B. Unbranched α (1→6)
C. Branched α (1→4)
D. Unbranched β (1→4)
Answer: D
10. C₁–C₄ glycosidic bond is not found in:
A. Maltose
B. Sucrose
C. Lactose
D. Starch
Answer: B
11. Glycosidic linkage in amylose is:
A. C₁–C₆ α
B. C₁–C₄ α
C. C₁–C₆ β
D. C₁–C₄ β
Answer: B
12. Glycosidic linkage in sucrose is between:
A. C₁ β-glucose & C₄ α-glucose
B. C₁ β-glucose & C₄ β-fructose
C. C₁ α-glucose & C₄ α-glucose
D. C₁ α-glucose & C₂ β-fructose
Answer: D
13. Which gives positive Fehling’s test?
A. Sucrose
B. Glucose
C. Fats
D. Protein
Answer: B
14. Glycogen is:
A. Polymer of β-D glucose
B. Structural polysaccharide
C. Similar to amylose
D. Highly branched like amylopectin
Answer: D
15. Incorrect statement is:
A. Aldose/ketose do not isomerise in alkaline medium
B. Carbohydrates are optically active
C. Pentaacetate does not react with hydroxylamine
D. Lactose has proper glycosidic linkage
Answer: A
16. Glucose + HI/red phosphorus gives:
A. n-hexane
B. n-heptane
C. n-pentane
D. n-octane
Answer: A
17. Sucrose is non-reducing because:
A. No free aldehyde/keto group
B. Contains fructose
C. Optically active
D. Stable
Answer: A
18. Saccharin is prepared from:
A. Toluene
B. Cyclohexane
C. Starch
D. Cellulose
Answer: A
19. α-maltose consists of:
A. α + β glucose (1→2)
B. α + α (1→2)
C. β + β (1→4)
D. α + α (1→4)
Answer: D
20. Least iodine value is:
A. Sunflower oil
B. Ginger oil
C. Groundnut oil
D. Ghee
Answer: D
21. Diabetic person carries glucose because:
A. Reduces sugar slowly
B. Increases slowly
C. Reduces instantly
D. Increases instantly
Answer: D
22. Chirality of DNA/RNA is due to:
A. D-sugar
B. L-sugar
C. Bases
D. Phosphate
Answer: A
23. Artificial sweetener with chlorine:
A. Aspartame
B. Saccharin
C. Sucralose
D. Alitame
Answer: C
24. Identify carbohydrate type (image-based):
A. 1,3,6
B. 1,3,4
C. 1,3,5
D. 2,3,5
Answer: A
25. α and β glucose are:
A. Enantiomers
B. Diastereomers
C. Anomers
D. C-5 isomers
Answer: C
26. Incorrect statement:
A. Fructose is furanose
B. Glucose is pyranose
C. Sucrose has peptide bond
D. Maltose is reducing
Answer: C
27. Oxyhaemoglobin stability difference:
A. 50
B. 200
C. 500
D. 300
Answer: D
28. Enzyme matching (correct set):
A. A–III, B–IV, C–I, D–V, E–II
B. A–III, B–IV, C–II, D–V, E–I
C. A–III, B–I, C–II, D–V, E–IV
D. A–III, B–I, C–II, D–IV, E–V
Answer: A
29. “D” in D-carbohydrates represents:
A. Dextrorotatory
B. Direct synthesis
C. Configuration
D. Optical activity
Answer: C
30. Invert sugar is:
A. Levorotatory
B. Dextrorotatory
C. Dextrorotatory
D. Optically inactive
Answer: A
Conclusion on Disaccharides Polysaccharides MCQs Advanced Level
Finally, the importance of Disaccharides polysaccharides mcqs advanced level cannot be overstated. They provide a comprehensive approach to learning, combining theory, application, and exam strategy.
In conclusion, consistent practice of Disaccharides polysaccharides mcqs advanced level is one of the most effective ways to master carbohydrate chemistry and excel in competitive exams.
