- Keneitsino Lydia
- March 20, 2026
30 Aniline vs Phenylamine MCQs with Explanation – Best Ultimate Questions for NEET, JEE & CUET to Boost Your Score
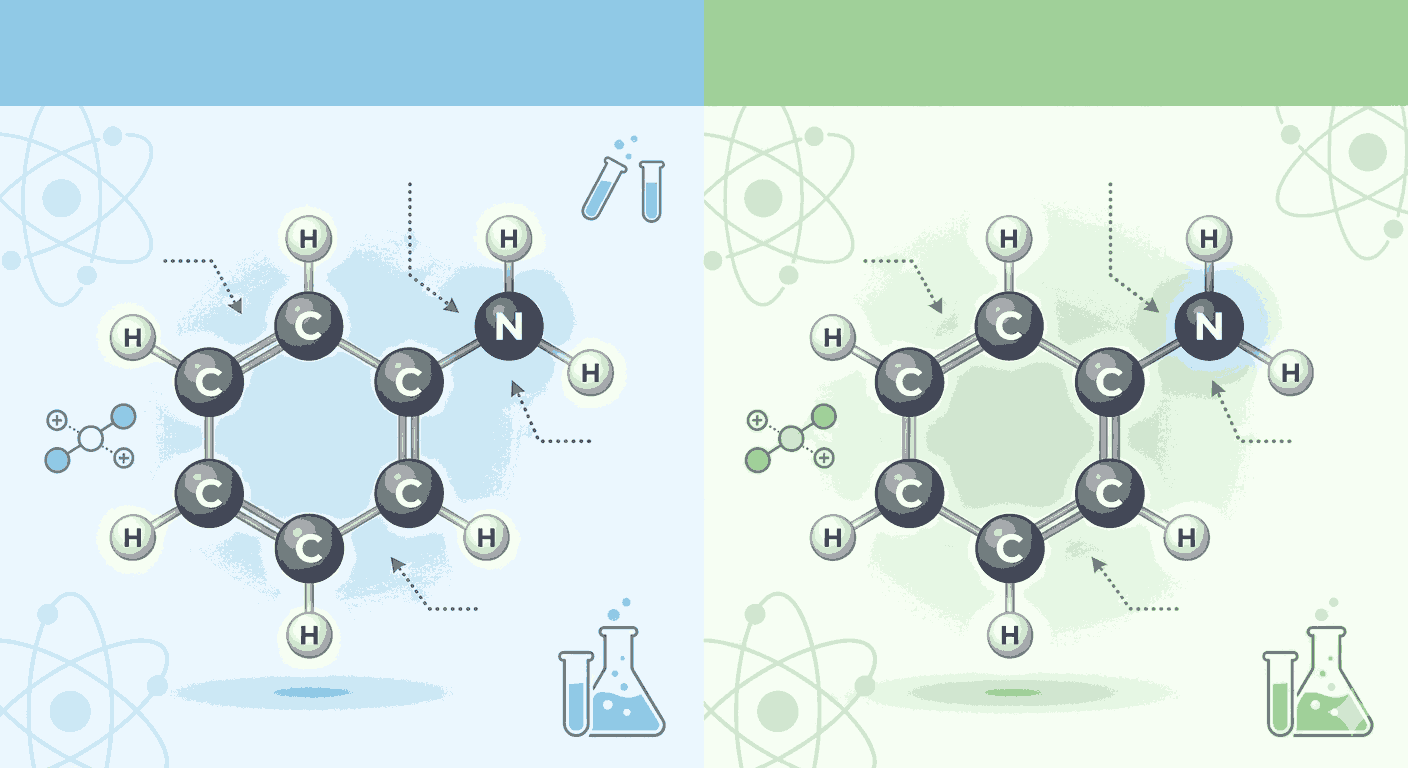
Aniline and phenylamine are two terms that often confuse aspirants studying organic chemistry in Class 12. In reality, both refer to the same compound, C₆H₅NH₂, but the difference lies in nomenclature and context of usage. Understanding this distinction becomes much easier through consistent practice using Aniline vs Phenylamine MCQs with explanation, which helps aspirants grasp both naming conventions and conceptual clarity. For exam preparation, especially NEET and JEE, mastering such terminology is essential, and Aniline vs Phenylamine MCQs with explanation play a crucial role in reinforcing these ideas.
Aniline is the common name, while phenylamine is the IUPAC-based systematic name derived from the phenyl group attached to an amine functional group. Aspirants often encounter both names interchangeably in textbooks and exams, making Aniline vs Phenylamine MCQs with explanation a valuable resource to avoid confusion. By repeatedly practicing Aniline vs Phenylamine MCQs with explanation, learners can confidently recognize that both names refer to the same aromatic amine.
The chemical structure of aniline consists of a benzene ring attached directly to an amino group (–NH₂). This structure significantly influences its properties, especially basicity and reactivity. Through Aniline vs Phenylamine MCQs with explanation, aspirants learn how resonance in the benzene ring affects the availability of the lone pair on nitrogen. This delocalization reduces the basic strength of aniline compared to aliphatic amines.
Another important concept highlighted in Aniline vs Phenylamine MCQs with explanation is the comparison of basicity between aniline and other amines. Because the lone pair on nitrogen participates in resonance with the aromatic ring, it becomes less available for protonation. This makes aniline a weaker base than ammonia and much weaker than aliphatic amines. Practicing Aniline vs Phenylamine MCQs with explanation helps aspirants internalize this key concept.
Reactivity patterns also differ significantly due to the presence of the amino group on the benzene ring. The –NH₂ group is an activating, ortho-para directing group in electrophilic substitution reactions. However, under strongly acidic conditions, it forms anilinium ion, which is deactivating. These nuanced concepts are best understood through Aniline vs Phenylamine MCQs with explanation, which present multiple scenarios and conditions.
In laboratory chemistry, aniline undergoes several characteristic reactions such as diazotization, bromination, and acetylation. Each of these reactions has specific conditions and products that are frequently tested in exams. By solving Aniline vs Phenylamine MCQs with explanation, aspirants can learn how these reactions proceed and what products are expected under different conditions.
Another area where Aniline vs Phenylamine MCQs with explanation are extremely helpful is distinguishing aniline from other amines using qualitative tests. For example, the Carbylamine test confirms the presence of primary amines, while the Hinsberg test helps classify amines. These practical aspects are often included in MCQs, making Aniline vs Phenylamine MCQs with explanation highly useful for exam-oriented preparation.
Moreover, Aniline vs Phenylamine MCQs with explanation improve analytical thinking by exposing aspirants to assertion-reason questions, conceptual comparisons, and reaction-based problems. This not only strengthens understanding but also enhances problem-solving speed, which is critical in competitive exams.
Regular revision using Aniline vs Phenylamine MCQs with explanation also helps aspirants retain important concepts without revisiting lengthy notes. It serves as a quick and efficient way to revise key points such as resonance, basicity, and reaction mechanisms.
Aspirants can also identify common mistakes through Aniline vs Phenylamine MCQs with explanation, such as assuming aniline behaves exactly like aliphatic amines. Recognizing and correcting these misconceptions improves accuracy and confidence.
In addition, Aniline vs Phenylamine MCQs with explanation provide exposure to different question patterns, helping aspirants adapt to various exam formats. This prepares them thoroughly for both board exams and entrance tests.
Aniline vs Phenylamine MCQs with explanation:
1. The correct order of boiling points of isomeric amines is:
a) Tertiary > Primary > Secondary
b) Secondary > Tertiary > Primary
c) Primary > Tertiary > Secondary
d) Primary > Secondary > Tertiary
Answer: d
2. Hoffmann bromamide reaction is used to convert:
a) Amine → Amide
b) Amide → Amine
c) Alcohol → Acid
d) Acid → Alcohol
Answer: b
3. Identify product C in the reaction:
Aniline → (CH₃CO)₂O/Pyridine → A → Br₂/CH₃COOH → B → Hydrolysis → C
a) Acetanilide
b) p-Bromoacetanilide
c) p-Bromoaniline
d) o-Bromoaniline
Answer: c
4. Nitration of aniline in acidic medium gives m-product because:
a) Nitro always goes meta
b) NH₂ is meta-directing
c) No substituent effect
d) Aniline forms anilinium ion
Answer: d
5. N,N-dimethyl aniline + NaNO₂/HCl gives:
a) p-Nitroso-N,N-dimethylaniline
b) m-Nitro derivative
c) Diazonium salt
d) N-nitroso compound
Answer: a
6. Aniline is used in manufacture of:
a) Aspirin
b) Cinnamic acid
c) Acetanilide
d) Salicylic acid
Answer: c
7.Assertion (A): Pyridine is more basic than pyrrole.
Reason (R): In pyrrole, the lone pair of electrons on nitrogen is involved in aromatic sextet, whereas in pyridine it is not.
a) Both A and R are correct and R is the correct explanation of A
b) Both A and R are correct but R is not the correct explanation
c) A is correct but R is incorrect
d) Both A and R are incorrect
Answer: a
8.Assertion (A): N,N-diethylethanamine is more basic than N,N-dimethylmethanamine.
Reason (R): Ethyl group shows stronger +I effect than methyl group.
a) Both A and R are correct and R is the correct explanation
b) Both A and R are correct but R is not the correct explanation
c) A is correct but R is incorrect
d) Both A and R are incorrect
Answer: a
9. Amine giving clear solution with Hinsberg reagent:
a) C₂H₅NH₂
b) (C₂H₅)₂NH
c) (C₂H₅)₃N
d) CH₃NHC₂H₅
Answer: a
10. Reaction converting acetamide to methanamine:
a) Hoffmann reaction
b) Stephen’s reaction
c) Gabriel synthesis
d) Carbylamine reaction
Answer: a
11. Compound giving foul smell with CHCl₃ + KOH:
a) PhCONH₂
b) PhNHCH₃
c) PhNH₂
d) PhOH
Answer: c
12. Hoffmann bromamide reaction is used to:
a) Prepare tertiary amine
b) Prepare all amines
c) Increase carbon chain
d) Decrease carbon chain
Answer: d
13. Hydrolysis of aromatic amide gives:
a) Acids
b) Amines
c) Alcohols
d) None
Answer: a
14. Reagent NOT used in Gabriel synthesis:
a) KOH
b) NaOH
c) HCl
d) Alkyl halide
Answer: c
15.Assertion (A): Aniline does not undergo Friedel–Crafts reaction easily.
Reason (R): The –NH₂ group reacts with AlCl₃ forming a complex, reducing ring activation.
a) Both A and R are correct and R is the correct explanation
b) Both A and R are correct but R is not the correct explanation
c) A is correct but R is incorrect
d) Both A and R are incorrect
Answer: a
16.Assertion (A): Aniline is a better nucleophile than anilinium ion.
Reason (R): Anilinium ion carries a positive charge on nitrogen.
a) Both A and R are correct and R is the correct explanation
b) Both A and R are correct but R is not the correct explanation
c) A is correct but R is incorrect
d) Both A and R are incorrect
Answer: a
17. Acetamide vs ethylamine test:
a) Water
b) KMnO₄
c) HCl
d) NaOH + heat
Answer: d
18.Which of the following orders of basic strength of amines is incorrect?
a) Aliphatic amines > Ammonia > Aromatic amines
b) Aromatic amines > Aliphatic amines > Ammonia
c) Secondary amines > Primary amines > Tertiary amines
d) Alkyl amines > Aniline
Answer: b
19. Most reactive towards electrophilic substitution:
a) Aniline
b) Nitrobenzene
c) Aniline HCl
d) Acetanilide
Answer: a
20. Which is a tertiary amine?
a) 1-Methylcyclohexylamine
b) Triethylamine
c) Tert-butylamine
d) N-methylaniline
Answer: b
21. Best reagent for conversion of amide to amine:
a) H₂
b) Br₂/NaOH
c) I₂/red P
d) LiAlH₄
Answer: d
22. Compound releasing N₂ with HNO₂:
a) Nitroethane
b) Triethylamine
c) Diethylamine
d) Ethylamine
Answer: d
23. Amine giving yellow oily liquid with HNO₂:
a) Triethylamine
b) Trimethylamine
c) Aniline
d) Methylphenylamine
Answer: d
24. Order of basicity:
a) 1 > 2 > 3 > 4
b) 1 > 3 > 2 > 4
c) 2 < 1 < 3 < 4
d) 4 > 3 > 2 > 1
Answer: c
25. Example of carbylamine reaction:
a) CS₂ reaction
b) HNO₂ reaction
c) CHCl₃ + KOH → isocyanide
d) Nitroso formation
Answer: c
26. Hinsberg test result (insoluble in alkali):
a) Primary amine
b) Secondary amine
c) Tertiary amine
d) Isocyanide
Answer: b
27.Assertion (A): Acylation of amines generally gives monosubstituted products.
Reason (R): Acyl group reduces nucleophilicity of nitrogen and prevents further substitution.
a) Both A and R are correct and R is the correct explanation
b) Both A and R are correct but R is not the correct explanation
c) A is correct but R is incorrect
d) Both A and R are incorrect
Answer: a
28. Compound not undergoing azo coupling:
a) Aniline
b) Phenol
c) Anisole
d) Nitrobenzene
Answer: d
29. Compound not identified by carbylamine test:
a) CHCl₃
b) C₆H₅-NH-C₆H₅
c) C₆H₅NH₂
d) CH₃CH₂NH₂
Answer: b
30. NaOH and Br₂ required in Hoffmann reaction:
a) 4 NaOH, 2 Br₂
b) 2 NaOH, 2 Br₂
c) 4 NaOH, 1 Br₂
d) 1 NaOH, 1 Br₂
Answer: c
Conclusion on Aniline vs Phenylamine MCQs with Explanation
Overall, understanding aniline and phenylamine is essential for mastering aromatic amines. Through consistent practice of Aniline vs Phenylamine MCQs with explanation, aspirants can build a strong conceptual foundation, improve their problem-solving abilities, and perform confidently in exams. Ultimately, Aniline vs Phenylamine MCQs with explanation serve as an effective and reliable tool for achieving success in chemistry.
