- Keneitsino Lydia
- March 25, 2026
Master Epimer and Anomer MCQs with Explanation Chemistry: Ultimate Success Guide
In the study of carbohydrates, understanding stereochemistry is extremely important for aspirants preparing for competitive exams. One of the most frequently tested concepts is the difference between epimers and anomers. This is why practicing epimer and anomer MCQs with explanation chemistry becomes essential for building a strong conceptual foundation.
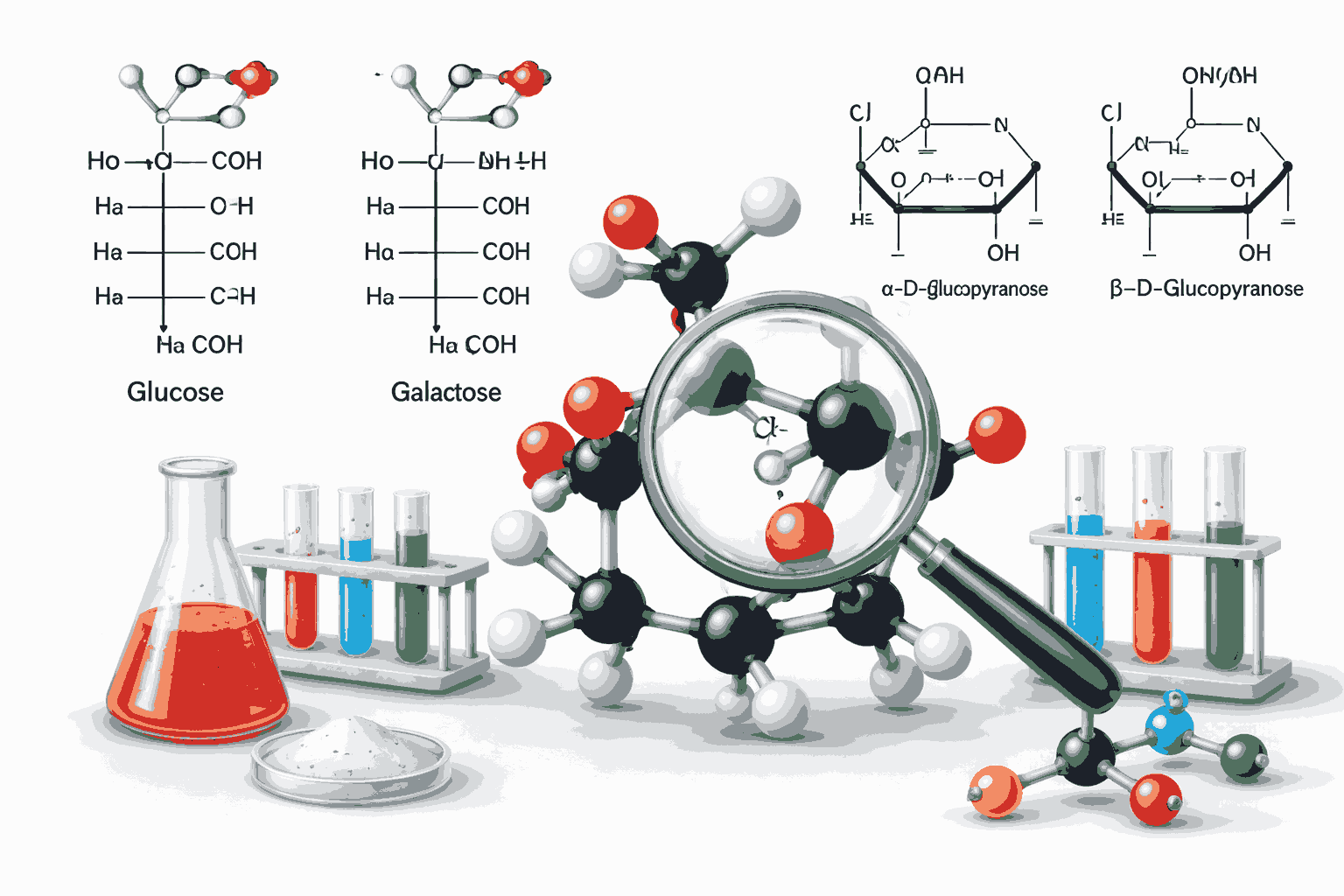
To begin with, epimers are stereoisomers that differ in configuration at only one specific chiral carbon atom. When aspirants solve epimer and anomer MCQs with explanation chemistry, they often encounter examples like glucose and galactose, which differ at the C-4 position. These subtle differences play a significant role in determining chemical properties and biological functions.
On the other hand, anomers are a special type of epimers that differ specifically at the anomeric carbon atom. In cyclic sugars like glucose, the anomeric carbon is the carbon derived from the carbonyl group. Understanding this distinction is crucial while solving epimer and anomer MCQs with explanation chemistry because it helps avoid common confusion between the two terms.
Another important aspect covered in epimer and anomer MCQs with explanation chemistry is the concept of mutarotation. Mutarotation refers to the change in optical rotation due to the interconversion between α and β forms of a sugar in solution. This phenomenon is directly related to anomers and is frequently tested in epimer and anomer MCQs with explanation chemistry.
Aspirants should also focus on structural representation while practicing epimer and anomer MCQs with explanation chemistry. Fischer and Haworth projections are commonly used to depict stereochemistry in carbohydrates. Being able to convert between these representations is an important skill that enhances performance in epimer and anomer MCQs with explanation chemistry.
One of the common mistakes aspirants make while solving epimer and anomer MCQs with explanation chemistry is confusing D- and L- configurations with epimers. It is important to note that D and L configurations refer to the orientation of the hydroxyl group on the penultimate carbon, whereas epimers differ at any one chiral center except the anomeric carbon.
Additionally, epimer and anomer MCQs with explanation chemistry often include questions about biological relevance. For example, glucose and galactose are epimers, but they are metabolized differently in the human body. Such conceptual understanding is essential when solving epimer and anomer MCQs with explanation chemistry in exams like NEET, JEE, and CUET.
The classification of carbohydrates is another topic closely linked with epimer and anomer MCQs with explanation chemistry. Monosaccharides, disaccharides, and polysaccharides all exhibit stereochemical variations. Recognizing these variations helps aspirants accurately answer epimer and anomer MCQs with explanation chemistry.
Furthermore, cyclic structures of sugars introduce the concept of hemiacetal and hemiketal formation, which leads to the formation of anomers. This concept is often emphasized in epimer and anomer MCQs with explanation chemistry, making it necessary for aspirants to understand ring formation mechanisms.
Another important point is that anomers can interconvert in solution, whereas epimers generally do not interconvert easily. This difference is a key concept frequently tested in epimer and anomer MCQs with explanation chemistry and should be clearly understood.
Practicing regularly with epimer and anomer MCQs with explanation chemistry helps aspirants develop problem-solving skills and improve their accuracy. It also enables them to identify patterns in questions and apply concepts effectively during exams.
In addition, many questions in epimer and anomer MCQs with explanation chemistry are application-based rather than direct theory. This meansaspirants must analyze structures and identify differences rather than rely solely on memorization.
Time management is another benefit of practicing epimer and anomer MCQs with explanation chemistry. As aspirants become familiar with different question types, they can solve them more quickly and confidently in actual exams.
Finally, revision plays a crucial role. Revisiting concepts through epimer and anomer MCQs with explanation chemistry ensures that aspirants retain information for a longer period and perform better under exam conditions.
Epimer and Anomer MCQs with Explanation Chemistry:
1. Assertion–Reason
Statement-I: Despite having an aldehyde group, glucose does not give Schiff’s test.
Statement-II: Glucose exists in α and β crystalline forms.
a) Both statements I and II are correct
b) Both statements I and II are incorrect
c) Statement I is correct but statement II is incorrect
d) Statement I is incorrect but statement II is correct
Answer: a
2. Carbohydrate storage forms
Carbohydrates are stored in plants and animals respectively as:
a) Glycogen, Starch
b) Glycogen, Glycogen
c) Starch, Starch
d) Starch, Glycogen
Answer: d
3. Reaction of glucose with HI
The major product of the reaction:
Glucose→HI,Δ→Glucose \xrightarrow{HI,\Delta} \rightarrow (high pressure conditions)
a) Cyclohexane
b) Benzene
c) Cyclohexaldehyde
d) Hexane
Answer: b
4. RNA type
Which of the following is NOT a type of RNA?
a) m-RNA
b) d-RNA
c) t-RNA
d) r-RNA
Answer: b
5. Assertion–Reason
Assertion: Glucose and fructose have the same D-configuration.
Reason: Both are dextrorotatory.
a) Both true, correct explanation
b) Both true, not explanation
c) Assertion true, Reason false
d) Assertion false, Reason true
Answer: c
6. Hydrolysis of sucrose
Hydrolysis of sucrose gives:
a) Dextrorotatory fructose & laevorotatory glucose
b) Both dextrorotatory
c) Both laevorotatory
d) Opposite incorrect combination
Answer: a
7. Type of fructose
Fructose is:
a) Ketohexose
b) Aldohexose
c) Ketopentose
d) Aldopentose
Answer: a
8. Ratio of products
Sucrose hydrolysis produces glucose and fructose in ratio:
a) 1 : 1
b) 2 : 1
c) 3 : 1
d) 4 : 1
Answer: a
9. Reducing sugars
Which are reducing sugars?
(i) Glucose (ii) Galactose (iii) Ribose (iv) Fructose
a) iii only
b) i, ii, iv
c) i, iii, iv
d) All
Answer: d
10. Composition of lactose
Lactose is composed of:
a) α-D-glucose only
b) α-D-glucose + β-D-glucose
c) β-D-galactose + β-D-glucose
d) α-D-galactose + β-D-glucose
Answer: c
11. Fermentation enzyme
Enzyme converting glucose → ethanol:
a) Invertase
b) Maltase
c) Diastase
d) Zymase
Answer: d
12. Tollen’s reagent reduction
Which reduces Tollen’s reagent?
a) Sucrose
b) Starch
c) Glucose
d) All
Answer: c
13. Carbohydrate statements
Correct statements:
a) (i) & (ii)
b) (ii) & (iii)
c) (ii), (iii), (iv)
d) (ii) & (iv)
Answer: b
14. When glucose reacts with H+(acidic medium), it mainly undergoes:
a) Oxidation
b) Reduction
c) Ring opening and closing(mutarotation)
d) Polymerizzation
Answer: c
15. Lactose statement
Incorrect statement about β-lactose:
a) Gives glucose + galactose
b) β-glycoside
c) Does not show mutarotation
d) Has 8 –OH groups
Answer: c
16. Tollens positive pair
a) Glucose, sucrose
b) Glucose, fructose
c) Hexanal, acetophenone
d) Fructose, sucrose
Answer: b
17. Glycerol reaction
Glycerol + oxalic acid →
a) Formic acid
b) Allyl alcohol
c) CO₂ + CO
d) Acrolein
Answer: a
18. Incorrect pair
a) Preservative – Sodium benzoate
b) Sweetener – Saccharin
c) Analgesic – Aspirin
d) Antacid – Iodoform
Answer: d
19. Assertion(A): Fructose is a reducing sugar.
Reason (R): Fructose can tautomerize to glucose in alkaline medium.
a) Both A and R are true , and R is correct explanation
b) Both A and R are true, but R is not correct explanation
c) A is true,R is false
d) A is false, R is true
Answer: a
20. Nature of sugars
Sugars are:
a) Sweet
b) Crystalline
c) Water soluble
d) All
Answer: d
21. Raffinose hydrolysis
Produces:
a) Glucose
b) Galactose
c) Fructose
d) All
Answer: d
22. IUPAC name of glucose
a) Pentahydroxy hexanal (wrong form)
b) Incorrect numbering
c) 2,3,4,5,6-pentahydroxy hexanal
d) Pentahydroxy hexanone
Answer: c
23. Monosaccharides
All monosaccharides ______ Tollen’s reagent.
a) Oxidize
b) Condense
c) Reduce
d) Add
Answer: c
24. Chiral centers in glucose
a) 4
b) 3
c) 2
d) 1
Answer: a
25. Property not due to H-bonding
a) Viscosity
b) Hygroscopic nature
c) Sweet taste
d) High boiling point
Answer: c
26. Charring reaction
Type of reaction:
a) Dehydration
b) Hydrolysis
c) Addition
d) Disproportionation
Answer: a
27. Which of the following artificial sweeteners is not stable on heating and therefore not suitable for cooking?
a) Saccharin
b) Aspartame
c) Sucrarlose
d) Alitame
Answer: b
28. Mutarotation causes change in :
a) Boiling point
b) Optical rotation
c) Molecular weight
d)Color
Answer: b
29. Incorrect glucose statement
a) Shows mutarotation
b) Has 4 chiral centers
c) Gives saccharic acid with Tollen’s reagent
d) Reacts with hydroxylamine
Answer: c
30. Yeast nutrition
Chemicals used:
a) (NH₄)₂SO₄, Na₃PO₄
b) (NH₄)₂SO₄, (NH₄)₃PO₄
c) Na₂SO₄, (NH₄)₃PO₄
d) Na₂SO₃, Na₃PO₄
Answer: b
Conclusion on Epimer and Anomer MCQs with Explanation Chemistry
In conclusion, mastering stereochemistry through epimer and anomer MCQs with explanation chemistry is essential for scoring well in chemistry exams. With consistent practice and a clear understanding of concepts, aspirants can confidently tackle even the most challenging questions related to epimers and anomers.
