- Keneitsino Lydia
- March 14, 2026
Best Physisorption vs Chemisorption Practice MCQs for Class 12 – Master Surface Chemistry Fast
Surface chemistry is an important chapter in Class 12 chemistry, and one of the most frequently tested topics within it is the difference between physical adsorption and chemical adsorption. Understanding these two types of adsorption is essential for aspirants preparing for board exams and competitive entrance tests. Practicing Physisorption vs Chemisorption Practice MCQs for Class 12 helps aspirants clearly distinguish between the two processes and strengthens conceptual understanding. Many examination questions focus on this comparison, which is why Physisorption vs Chemisorption Practice MCQs for Class 12 are highly useful for revision.
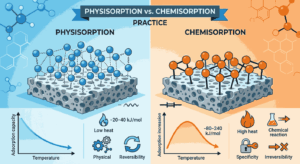
Physisorption, also known as physical adsorption, occurs when gas molecules attach to the surface of a solid due to weak van der Waals forces. These forces are relatively weak, so the adsorption process is usually reversible. On the other hand, chemisorption involves the formation of strong chemical bonds between the adsorbate and the adsorbent surface. The differences between these two mechanisms are frequently explored in Physisorption vs Chemisorption Practice MCQs for Class 12, allowing aspirants to understand how each process behaves under different conditions. Regular practice with Physisorption vs Chemisorption Practice MCQs for Class 12 helps reinforce these important distinctions.
One of the main differences between physisorption and chemisorption is the strength of the forces involved. Physisorption relies on weak intermolecular attractions, while chemisorption involves strong chemical bonding. Because of this difference, the heat of adsorption in chemisorption is much higher than in physisorption. This concept is commonly tested in Physisorption vs Chemisorption Practice MCQs for Class 12, making it essential for aspirants to remember. When aspirants repeatedly solve Physisorption vs Chemisorption Practice MCQs for Class 12, they become more confident in identifying the thermodynamic properties associated with each type.
Temperature also affects physisorption and chemisorption differently. Physisorption generally occurs more readily at lower temperatures because increased thermal energy can overcome the weak van der Waals forces holding the molecules to the surface. Chemisorption, however, may require higher temperatures because activation energy is needed to form chemical bonds. These temperature effects are often highlighted in Physisorption vs Chemisorption Practice MCQs for Class 12, helping aspirants understand how adsorption changes under varying thermal conditions. By reviewing Physisorption vs Chemisorption Practice MCQs for Class 12, aspirants can easily remember which process is favored at different temperatures.
Another key difference lies in the number of layers formed on the adsorbent surface. Physisorption can form multiple layers of adsorbed molecules because the forces involved are weak and allow additional layers to accumulate. Chemisorption, in contrast, usually forms only a single molecular layer since the chemical bonds occupy specific active sites on the surface. This property is frequently examined in Physisorption vs Chemisorption Practice MCQs for Class 12, making it a crucial point of comparison. Continuous practice with Physisorption vs Chemisorption Practice MCQs for Class 12 helps aspirants quickly recognize these structural differences.
The specificity of adsorption also varies between the two types. Physisorption is generally non-specific, meaning that many different gases can be adsorbed by the same adsorbent. Chemisorption, however, is highly specific because it depends on the chemical reactivity between the adsorbate and the surface. Questions related to specificity are common in Physisorption vs Chemisorption Practice MCQs for Class 12, as they test a deeper understanding of surface chemistry. Through repeated exposure to Physisorption vs Chemisorption Practice MCQs for Class 12, aspirants learn to identify which adsorption process is involved in a given reaction.
Another factor that influences adsorption is pressure. Increasing pressure generally increases the extent of adsorption in both physisorption and chemisorption, but the effect is more pronounced in physical adsorption because additional layers can form. This relationship between pressure and adsorption is frequently discussed in Physisorption vs Chemisorption Practice MCQs for Class 12. By solving Physisorption vs Chemisorption Practice MCQs for Class 12, aspirants gain confidence in analyzing pressure-dependent adsorption behavior.
The comparison between physisorption and chemisorption is not only important for exams but also for understanding industrial and practical applications. Adsorption processes are widely used in catalysis, gas purification, and environmental chemistry. Knowing the differences between the two types of adsorption helps explain why certain catalysts work more effectively under specific conditions. These practical insights are often reinforced through Physisorption vs Chemisorption Practice MCQs for Class 12, which connect theoretical knowledge with real-world applications. Practicing Physisorption vs Chemisorption Practice MCQs for Class 12 therefore strengthens both conceptual understanding and practical awareness.
Q1. Which of the following statements is correct about oleum?
A. It is prepared by adsorption of SO₃ in conc. H₂SO₄
B. It contains O–O groups
C. It has six OH groups
D. None of these
Answer: A
Q2.
3.6 g of oxygen is adsorbed on 1.2 g of metal powder. What volume of oxygen is adsorbed per gram of adsorbent at 1 atm and 273 K?
A. 9.0 L g⁻¹
B. 1 L g⁻¹
C. 2.0 L g⁻¹
D. None of these
Answer: C
Q3. Correct matching between Item-I and Item-II
| Item I | Item II |
|---|---|
| Benzaldehyde | Adsorbate |
| Alumina | Adsorbent |
| Acetonitrile | Dynamic phase |
Options:
A. A–R, B–Q, C–P
B. None of these
C. A–P, B–Q, C–R
D. A–P, B–R, C–Q
Answer: A
Q4.
Based on the critical temperature data of gases (H₂, CH₄, CO₂, SO₂), which gas shows least adsorption on charcoal?
A. CH₄
B. SO₂
C. CO₂
D. H₂
Answer: D
Q5.
In adsorption of oxalic acid on activated charcoal, the activated charcoal acts as:
A. Adsorber
B. Absorbate
C. Adsorbent
D. Occlusion
Answer: C
Q6.
Adsorption of gases on solid surface is generally exothermic because:
A. Enthalpy is positive
B. Entropy decreases
C. Entropy increases
D. Free energy increases
Answer: B
Q7.
Which statement is true regarding chemisorption of a gas on a solid surface?
A. Adsorption increases with increase in temperature initially
B. No compound formation occurs
C. Weak Van der Waals forces operate
D. Multimolecular layers form
Answer: A
Q8.
Which statement is true about adsorption?
A. ΔH < 0 and ΔS < 0
B. ΔH > 0 and ΔS < 0
C. ΔH < 0 and ΔS > 0
D. ΔH = 0 and ΔS > 0
Answer: A
Q9.
Which statement is not applicable to chemisorption?
A. Highly specific
B. Slow process
C. Independent of temperature
D. Irreversible
Answer: C
Q10.
The rate of chemisorption of a gas:
A. Decreases with pressure
B. Increases with pressure
C. Independent of pressure
D. Independent of temperature
Answer: B
Q11. Select the incorrect statement.
A. Physical adsorption is reversible while chemical adsorption is irreversible
B. High pressure favors physical adsorption while low pressure favors chemical adsorption
C. Physical adsorption is non-specific
D. Chemical adsorption requires high activation energy
Answer: B
Q12.
Which statement is true?
A. Adsorption of H₂ by platinum black is sorption
B. Adsorption of H₂ by charcoal is occlusion
C. Adsorption of H₂ by platinum black is occlusion
D. Absorption of H₂ by charcoal is chemisorption
Answer: C
Q13.
If adsorption of a gas on metal surface is spontaneous and exothermic:
A. H increases
B. S increases
C. G increases
D. S decreases
Answer: D
Q14.
Which process does not use adsorption?
A. Froth flotation
B. Chromatography
C. Decolorization of sugar
D. Dissolution of sugar in water
Answer: D
Q15.
Which is not favorable for physical adsorption?
A. High pressure
B. High temperature
C. High critical temperature of gas
D. Low temperature
Answer: B
Q16.
For spontaneous adsorption of H₂ gas on solid surface:
A. Exothermic and ΔS < 0
B. Endothermic and ΔS > 0
C. Exothermic and ΔS > 0
D. Endothermic and ΔS < 0
Answer: A
Q17.
Properties of physical adsorption are:
(i) reversible
(ii) unimolecular layer formation
(iii) low heat of adsorption
(iv) occurs at low temperature
A. I, II, III
B. I, III, IV
C. II, III, IV
D. I, II, IV
Answer: B
Q18.
According to Langmuir isotherm, at very high pressure adsorption:
A. Reaches constant value
B. Increases indefinitely
C. Decreases
D. Increases then decreases
Answer: A
Q19.
In which property do physisorption and chemisorption resemble each other?
A. Force of attraction
B. Heat of adsorption
C. Temperature effect
D. Surface area effect
Answer: D
Q20.
3 g activated charcoal added to 50 mL of 0.06 N acetic acid. Final strength = 0.042 N.
Amount adsorbed per gram charcoal is:
A. 18 mg
B. 36 mg
C. 42 mg
D. 54 mg
Answer: A
Q21.
Which figure represents physical adsorption?
A. Diagram 1
B. Diagram 2
C. Diagram 3
D. Diagram 4
Answer: D
Q22.
Gold sol has:
A. solid–solid lyophilic
B. liquid–liquid lyophobic
C. solid–liquid lyophobic
D. solid–liquid lyophilic
Answer: C
Q23.
For adsorption at boiling point, thermodynamic properties at point C are:
A. ΔH = TΔS ; ΔG = 0
B. ΔH > TΔS ; ΔG positive
C. ΔH < TΔS ; ΔG negative
D. ΔH = TΔS = ΔG = 0
Answer: A
Q24.
Which statement is false during adsorption of gas on metal surface?
A. Enthalpy change positive
B. Entropy change negative
C. Both decrease
D. Free energy negative
Answer: A
Q25.
In which aspect do physisorption and chemisorption resemble?
A. Both are exothermic
B. Both multilayered
C. Both reversible
D. Both favored at high temperature
Answer: A
Q26.
Extent of physisorption increases with:
A. Increase temperature
B. Decrease surface area
C. Decrease pressure
D. Decrease temperature
Answer: D
Q27.
Which is not true for physical adsorption?
A. Reversible
B. Needs activation energy
C. Multilayer adsorption
D. Increases with pressure
Answer: B
Q28.
Assertion: Adsorption shown more by solids than liquids
Reason: Charcoal and silica act as good adsorbents
A. Both correct and reason explains
B. Both correct but reason not explanation
C. Assertion correct reason wrong
D. Assertion wrong reason correct
Answer: B
Q29.
Which is not true for adsorption of gas on solid?
A. Increase temperature favors adsorption
B. Enthalpy negative
C. Surface phenomenon
D. Entropy negative
Answer: A
Q30.
Which statement is incorrect?
A. Heat of physisorption < chemisorption
B. Milk is an emulsion
C. Physical adsorption increases with temperature
D. Smoke is aerosol
Answer: C
Conclusion on Physisorption vs Chemisorption Practice MCQs for Class 12
In conclusion, mastering the differences between physisorption and chemisorption is essential for aspirants studying surface chemistry in Class 12. These concepts involve thermodynamics, kinetics, and molecular interactions, making them both interesting and important. Regular practice through Physisorption vs Chemisorption Practice MCQs for Class 12 allows aspirants to revise the topic thoroughly and improve their accuracy in exams. By consistently reviewing Physisorption vs Chemisorption Practice MCQs for Class 12, aspirants can build a strong foundation in adsorption phenomena and perform confidently in both board and competitive examinations.
