- Keneitsino Lydia
- March 13, 2026
Powerful Freundlich Isotherm MCQs Class 12 with Answers for Smart Learning
Surface chemistry is an important part of Class 12 chemistry, and adsorption is one of its most scoring areas. Among adsorption topics, the Freundlich adsorption isotherm has special importance because it explains how the amount of gas adsorbed on a solid surface changes with pressure at constant temperature. For aspirants preparing for board exams and entrance tests, Freundlich Isotherm MCQs Class 12 with Answers is a highly useful practice area. By working through Freundlich Isotherm MCQs Class 12 with Answers, aspirants can strengthen both conceptual understanding and numerical confidence.
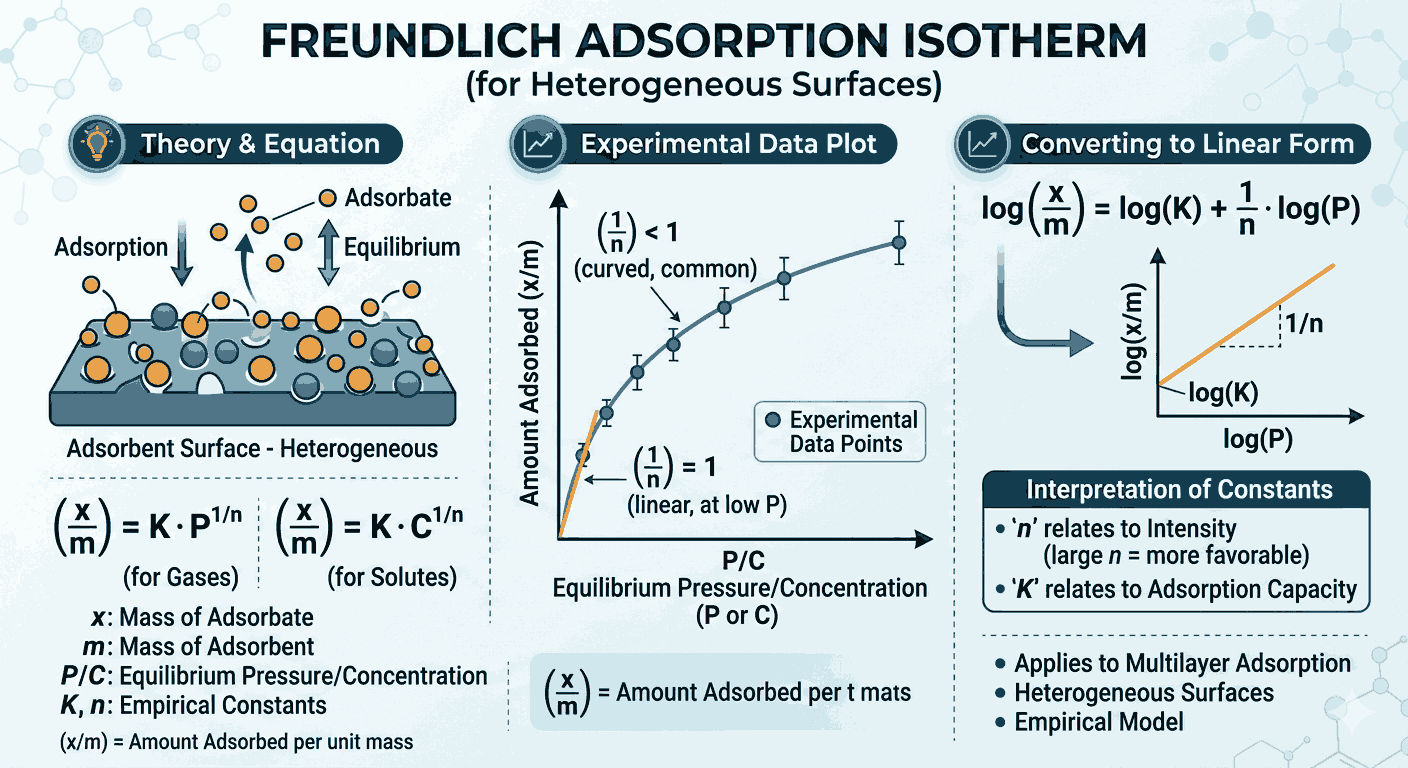
The Freundlich adsorption isotherm is an empirical relationship. It is used to describe adsorption when the amount of gas adsorbed per unit mass of adsorbent increases with pressure, but not in a simple linear way. In study materials, Freundlich Isotherm MCQs Class 12 with Answers often focus on this non-linear behavior, especially how adsorption changes at low, moderate, and high pressure. This makes Freundlich Isotherm MCQs Class 12 with Answers an essential revision topic in surface chemistry.
The standard mathematical form of the Freundlich isotherm is:
xm=kp1/n\frac{x}{m} = kp^{1/n}
Here, xx is the mass of gas adsorbed, mm is the mass of adsorbent, pp is the pressure, and kk and nn are constants for a given system at a fixed temperature. Many questions in Freundlich Isotherm MCQs Class 12 with Answers test whether aspirants can identify this equation correctly. In addition, Freundlich Isotherm MCQs Class 12 with Answers often check whether aspirants know that the logarithmic form gives a straight-line graph.
When the Freundlich equation is written in logarithmic form, it becomes:
logxm=logk+1nlogp\log \frac{x}{m} = \log k + \frac{1}{n} \log p
This form is very important because it shows that a plot of log(x/m)\log(x/m) versus logp\log p is a straight line. The slope of that line is 1/n1/n, and the intercept is logk\log k. This graphical interpretation is one of the most frequently tested ideas in Freundlich Isotherm MCQs Class 12 with Answers. In fact, many aspirants improve graph-based understanding mainly through repeated practice of Freundlich Isotherm MCQs Class 12 with Answers.
Another important point is that the value of 1/n1/n generally lies between 0 and 1. This tells us that adsorption increases with pressure, but not too sharply. It also reflects the fact that adsorption becomes less efficient at very high pressure. Questions about the range and meaning of 1/n1/n appear regularly in Freundlich Isotherm MCQs Class 12 with Answers, so this concept should be remembered carefully. A strong grip on this point makes Freundlich Isotherm MCQs Class 12 with Answers much easier to solve.
At low pressure, adsorption increases almost proportionally with pressure. At moderate pressure, the Freundlich relation works well. However, at high pressure, the Freundlich isotherm fails because the surface of the adsorbent begins to approach saturation. This limitation is another favorite exam area, and it is commonly highlighted in Freundlich Isotherm MCQs Class 12 with Answers. Aspirants who revise the limitations properly usually perform better in Freundlich Isotherm MCQs Class 12 with Answers than those who only memorize the formula.
Freundlich adsorption isotherm is mainly associated with physical adsorption, though the equation is used more generally as an empirical relationship. Physical adsorption is usually reversible, favoured at low temperature, and depends on surface area. These connections between adsorption properties and isotherm behavior are commonly asked in Freundlich Isotherm MCQs Class 12 with Answers. That is why Freundlich Isotherm MCQs Class 12 with Answers should not be studied as isolated formula questions alone.
Aspirants should also know how pressure affects adsorption numerically. If pressure changes, the amount adsorbed changes according to the exponent 1/n1/n. This means doubling the pressure does not usually double adsorption. Such proportionality-based questions are common in Freundlich Isotherm MCQs Class 12 with Answers, especially in board-style numericals. Regular practice of Freundlich Isotherm MCQs Class 12 with Answers helps aspirants handle these calculations quickly.
Temperature also matters. Since physical adsorption is exothermic, lowering the temperature generally increases adsorption. This is another idea linked with Freundlich behavior in school-level chemistry. Therefore, some conceptual questions in Freundlich Isotherm MCQs Class 12 with Answers combine pressure, temperature, and adsorption amount in a single problem. Solving Freundlich Isotherm MCQs Class 12 with Answers improves the ability to connect all these variables together.
From an exam perspective, this topic is scoring because the same ideas repeat in different forms: identifying the correct equation, interpreting the graph, finding slope or intercept, deciding the valid range of 1/n1/n, and understanding why the isotherm fails at high pressure. These repeated patterns make Freundlich Isotherm MCQs Class 12 with Answers a smart practice set for last-minute revision. With enough exposure to Freundlich Isotherm MCQs Class 12 with Answers, aspirants become faster and more accurate.
Freundlich Isotherm MCQs Class 12 with Answers:
1. Wrong statement about catalysis
Q1. Which one of the following statements is wrong?
A. In homogeneous catalysis, reactants, products, and catalyst are in the same phase.
B. A catalyst accelerates the rate of reaction by bringing down the energy of activation.
C. A catalyst alters the equilibrium constant.
D. The mass of catalyst remains the same after reaction.
Answer: C
2. Slope in Freundlich plot
Q2. In a plot of log(xm)\log\left(\frac{x}{m}\right) vs logp\log p for the adsorption of a gas on a solid, a straight line is obtained. The slope of that straight line is:
A. 1n\frac{1}{n}
B. KK
C. nn
D. logK\log K
Answer: A
3. Freundlich adsorption isotherm equation
Q3. In the Freundlich isotherm equation, the term represents:
A.Pressure of the gas
B. Mass of adsorbent
C. Amount of gas adsorbed per unit mass of absorbent
D. Temperature of the system
Answer: C
4. Freundlich isotherm equation
Q4. Which one of the following equations represents Freundlich adsorption isotherm?
A. xm=kp\frac{x}{m} = kp
B. xm=kp1/n\frac{x}{m} = kp^{1/n}
C. logxm=kp1/n\log \frac{x}{m} = kp^{1/n}
D. logxm=kplogp\log \frac{x}{m} = k_p \log p
Answer: B
5. Proportionality from given graph
Q5. Adsorption of a gas follows Freundlich adsorption isotherm. If the plot of xm\frac{x}{m} versus logp\log p is as shown, then xm\frac{x}{m} is proportional to:
A. p2/3p^{2/3}
B. p3/2p^{3/2}
C. p2p^2
D. p3p^3
Answer: A
6. Logarithmic Freundlich form
Q6. Freundlich equation for adsorption of gases on a solid at constant temperature can be expressed as:
A. logxm=logP+1nlogK\log \frac{x}{m} = \log P + \frac{1}{n}\log K
B. logxm=logK+1nlogP\log \frac{x}{m} = \log K + \frac{1}{n}\log P
C. None of these
D. xm=logP+1nlogK\frac{x}{m} = \log P + \frac{1}{n}\log K
Answer: B
7. Incorrect property of physical adsorption
Q7. Which one of the following characteristics is not correct for physical adsorption?
A. Adsorption on solids is reversible.
B. Adsorption increases with increase in temperature.
C. Adsorption is spontaneous.
D. Both enthalpy and entropy of adsorption are negative.
Answer: B
8. Condition increasing physical adsorption
Q8. Rate of physical adsorption increases with:
A. Decrease in surface area
B. Decrease in temperature
C. Decrease in pressure
D. Increase in temperature
Answer: B
9. Correct statement according to adsorption isotherm
Q9. Which of the following is correct according to adsorption isotherm?
A. xm∝p0\frac{x}{m} \propto p^0
B. xm∝p1\frac{x}{m} \propto p^1
C. xm∝pp1/n\frac{x}{m} \propto \frac{p}{p^{1/n}}
D. All of these
Answer: D
10. Curves not following Freundlich isotherm
Q10. Among the following, the number of curves not in accordance with Freundlich adsorption isotherm is:
A. Option 1
B. Option 2
C. Option 3
D. Option 4
Answer: C
11. Assertion–Reason on soap micelles
Q11. Assertion (A): The aggregate of carboxylate ions of fatty acids forms a spherical shape.
Reason (R): Soap helps in emulsification and washing away of oils.
A. Both (A) and (R) are correct and (R) is the correct explanation of (A).
B. Both (A) and (R) are correct but (R) is not the correct explanation of (A).
C. (A) is correct but (R) is incorrect.
D. (A) is incorrect but (R) is correct.
Answer: B
12. Failure of Freundlich isotherm
Q12. Freundlich adsorption isotherm fails under which of the following conditions?
A. High temperature
B. Low temperature
C. High pressure
D. Low pressure
Answer: C
13. Chemisorption statement
Q13. Which one of the following statements is correct about chemisorption?
A. It is reversible and favoured at high temperature.
B. It is irreversible and favoured at high temperature.
C. It is reversible and favoured at low temperature.
D. It is irreversible and favoured at low temperature.
Answer: B
14. Correct statements about adsorption
Q14. Which statements among the following are correct?
(i) Freundlich isotherm fails at high pressure of the gas
(ii) ΔH<0\Delta H < 0 for both physical and chemical adsorption
(iii) Physical adsorption is non-selective
(iv) Chemical adsorption is reversible, whereas physical adsorption is irreversible
A. (i), (ii), (iii), (iv)
B. (i), (ii), (iii)
C. (i), (ii), (iv)
D. (i), (iii)
Answer: B
15. Value of n from pressure exponent
Q15. In Freundlich adsorption isotherm at moderate pressure, the extent of adsorption xm\frac{x}{m} is directly proportional to PxP^x. The value of nn is:
A. 1n\frac{1}{n}
B. 2
C. 0
D. Infinity
Answer: A
16. Value of n from CO₂ adsorption
Q16. CO₂ gas adsorbs on charcoal following Freundlich adsorption isotherm. For a given amount of charcoal, the mass of CO₂ adsorbed becomes 64 times when the pressure of CO₂ is doubled. The value of nn in the Freundlich equation is ____ × 10⁻².
A. 17
B. 14
C. 13
D. 9
Answer: A
17. Vitamin example
Q17. Which among the following acts as a vitamin?
A. Aspartic acid
B. Ascorbic acid
C. Saccharin acid
D. Adipic acid
Answer: B
18. Mass adsorbed from slope and intercept
Q18. For Freundlich adsorption isotherm, a plot of log(xm)\log\left(\frac{x}{m}\right) versus logP\log P gives a straight line. The intercept and slope are 0.4771 and 2, respectively. The mass of gas adsorbed per gram of adsorbent if the initial pressure is 0.04 atm is ____ × 10⁻⁴.
A. 48
B. 50
C. 52
D. 55
Answer: A
19. Correct statement when n = 5
Q19. For a gas-solid adsorbent system, the adsorption isotherm xm=KP1/n\frac{x}{m} = KP^{1/n} is applicable, where n=5n = 5. Point out the correct statement.
A. xm\frac{x}{m} vs PP is a linear graph with slope equal to 5.
B. logxm\log \frac{x}{m} vs logP\log P is a straight line with slope equal to 5.
C. logxm\log \frac{x}{m} vs logP\log P is a straight line with slope equal to 0.2.
D. None of these.
Answer: C
20. Conditions for increase in physical adsorption
Q20. A gas undergoes physical adsorption on a surface and follows the Freundlich adsorption isotherm equation xm=Kp0.5\frac{x}{m} = Kp^{0.5}. Adsorption of the gas increases with:
A. Increase in pressure and increase in temperature
B. Increase in pressure and decrease in temperature
C. Decrease in pressure and decrease in temperature
D. Decrease in pressure and increase in temperature
Answer: B
21. Proportionality from given plot
Q21. Adsorption of a gas follows Freundlich adsorption isotherm. In the given plot, xm\frac{x}{m} is proportional to:
A. p1/2p^{1/2}
B. p3/2p^{3/2}
C. p1/4p^{1/4}
D. p3p^{3}
Answer: A
22. Correct Freundlich form
Q22. The correct option representing a Freundlich adsorption isotherm is:
A. xm=kp0.3\frac{x}{m} = kp^{0.3}
B. xm=kp2.5\frac{x}{m} = kp^{2.5}
C. xm=kp−0.5\frac{x}{m} = kp^{-0.5}
D. xm=kp−1\frac{x}{m} = kp^{-1}
Answer: A
23. Proportion of adsorption at 4 bar
Q23. The values of slope and intercept in the graph of Freundlich adsorption isotherm at 25°C are 0.5 and 0.477 respectively. What will be the proportion of adsorption at 4 bar pressure?
A. 2
B. 4
C. 24
D. 6
Answer: D
24. Condition where x/m equals pressure
Q24. Under what condition will xm\frac{x}{m} be equal to the pressure of the gas on the surface of the adsorbent?
A. At low pressure
B. At high pressure
C. At low temperature
D. At high temperature
Answer: A
25. Correct statement for linear plot
Q25. For a linear plot of log(x/m)\log(x/m) versus logp\log p in a Freundlich adsorption isotherm, which of the following statements is correct?
A. 1/n1/n appears as the intercept.
B. Only 1/n1/n appears as the slope.
C. log(1/n)\log(1/n) appears as the intercept.
D. Both kk and 1/n1/n appear in the slope term.
Answer: B
26. Straight line for Freundlich isotherm
Q26. Which one of the following gives a straight line for Freundlich adsorption isotherm?
A. logxm\log \frac{x}{m} vs logp\log p
B. xm\frac{x}{m} vs 1p\frac{1}{p}
C. logxm\log \frac{x}{m} vs log1p\log \frac{1}{p}
D. xm\frac{x}{m} vs pp
Answer: A
27. Slope of AB line
Q27. In Freundlich adsorption isotherm, slope of AB line is:
A. nn, with n=0.1n = 0.1 to 0.5
B. logn\log n, with n>1n > 1
C. log1n\log \frac{1}{n}, with n<1n < 1
D. 1n\frac{1}{n}, with 1n=0\frac{1}{n} = 0 to 1
Answer: D
28. Value of x/m at 0.2 atm
Q28. The graph between log(xm)\log\left(\frac{x}{m}\right) and logp\log p is a straight line at angle 45° with intercept OA as shown. Hence, (xm)\left(\frac{x}{m}\right) at a pressure of 0.2 atm is:
A. 0.8
B. 0.6
C. 0.4
D. 0.2
Answer: C
29. Range of 1/n
Q29. What is the value of 1/n1/n in Freundlich adsorption isotherm?
A. Between 2 and 4 in all cases
B. Between 0 and 1 in all cases
C. 1 in case of chemisorption
D. 1 in case of physical adsorption
Answer: B
30. Nature of n in Freundlich equation
Q30. In the Freundlich adsorption isotherm equation
logxm=logk+1nlogp\log \frac{x}{m} = \log k + \frac{1}{n}\log p,
the value of nn is:
A. Any value from 0 to 1
B. A negative integer
C. A positive integer
D. A positive or a negative fractional number
Answer: C
Conclusion on Freundlich Isotherm MCQs Class 12 with Answers
In conclusion, the Freundlich adsorption isotherm is a compact but very important topic in Class 12 chemistry. It combines equation-based learning, graph interpretation, and conceptual understanding of adsorption. Because it appears frequently in exams, aspirants should revise it thoroughly and repeatedly. Focused practice through Freundlich Isotherm MCQs Class 12 with Answers can make the chapter easier, clearer, and more scoring. Consistent revision with Freundlich Isotherm MCQs Class 12 with Answers is one of the best ways to master this part of surface chemistry.
