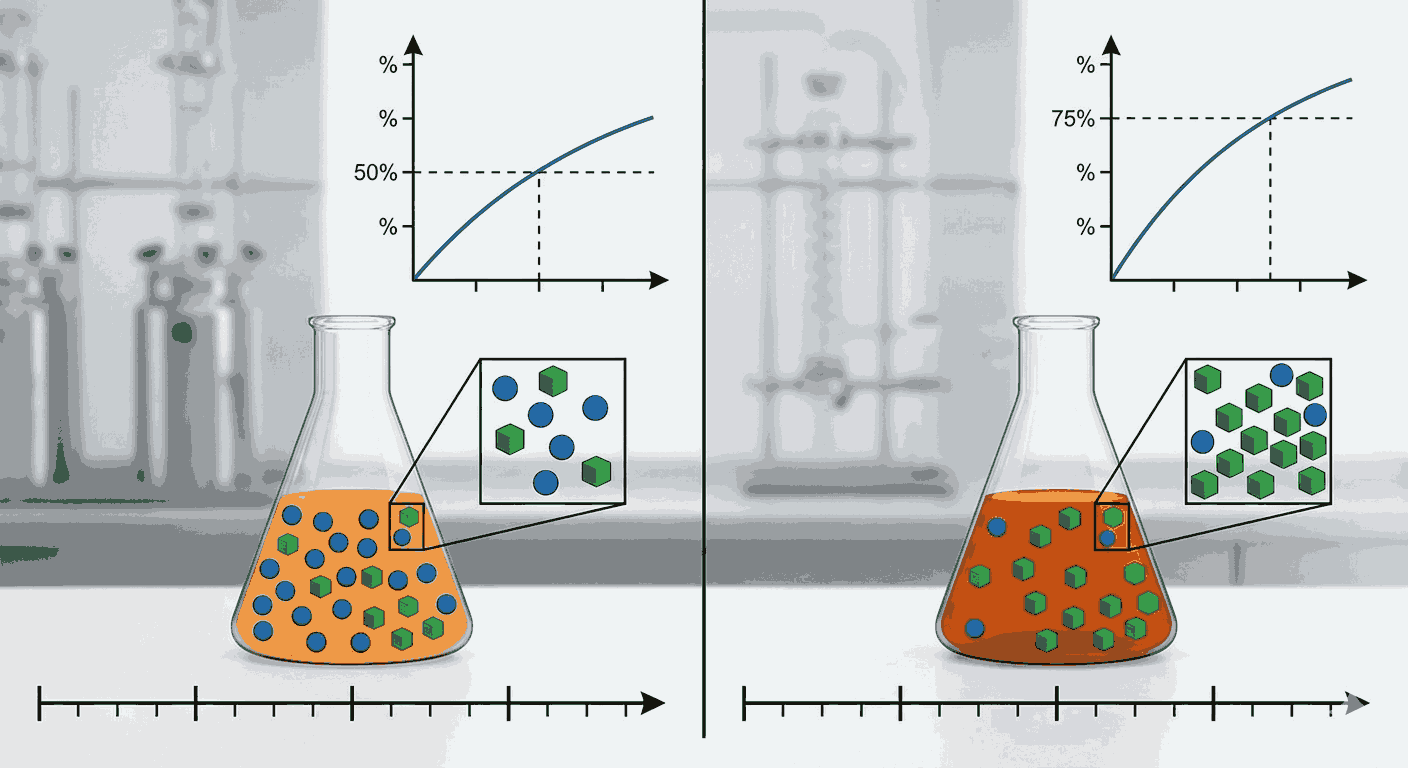
- Keneitsino Lydia
- March 12, 2026
Powerful 50 percent and 75 percent Completion MCQs in Kinetics for NEET, JEE & CUET with Answers
Chemical kinetics is an important branch of chemistry that studies the rate at which chemical reactions occur and the factors that influence those rates. Among the many numerical problems included in this topic, questions related to percentage completion of reactions are very common in examinations. Aspirants frequently encounter problems involving half-life, fractional completion, and time required for certain percentages of reactants to convert into products. Practicing 50 percent and 75 percent completion MCQs in kinetics helps learners develop a deeper understanding of how reaction rates and reaction time are related.
One of the most basic concepts in kinetics is the idea of reaction completion percentage. When we say a reaction is 50 percent complete, it means half of the initial reactant has been converted into products. Similarly, when a reaction reaches 75 percent completion, three-fourths of the initial reactant has reacted. These types of numerical concepts are often tested through 50 percent and 75 percent completion MCQs in kinetics, making them very important for aspirants preparing for board examinations and competitive tests.
In chemical kinetics, many reactions follow first-order behavior, where the rate of reaction depends on the concentration of a single reactant. For such reactions, the half-life plays a crucial role in determining how quickly a reaction proceeds. The half-life is defined as the time required for 50 percent of the reactant to be consumed. Understanding this concept is essential for solving 50 percent and 75 percent completion MCQs in kinetics, because these problems usually rely on the mathematical relationship between half-life and time.
For a first-order reaction, the half-life remains constant regardless of the initial concentration. This property makes calculations involving percentage completion much easier. For example, if the half-life of a reaction is known, aspirants can easily determine how much reactant remains after a certain number of half-lives. In many 50 percent and 75 percent completion MCQs in kinetics, the questions require aspirants to calculate the remaining concentration after multiple half-lives.
Another important idea is that the time required for 75 percent completion of a first-order reaction is greater than the half-life. This happens because reaching 75 percent completion means only 25 percent of the reactant remains. Aspirants solving 50 percent and 75 percent completion MCQs in kinetics often need to apply logarithmic relationships to determine the exact time required for this level of reaction progress.
The mathematical treatment of reaction completion often uses the integrated rate law for first-order reactions. This equation helps determine the concentration of reactant remaining after a certain time. When aspirants practice 50 percent and 75 percent completion MCQs in kinetics, they become more comfortable applying this formula to different numerical scenarios.
Understanding percentage completion also helps in interpreting reaction progress graphs. These graphs typically show how reactant concentration decreases with time. By analyzing the graph, aspirants can identify when a reaction reaches 50 percent completion or 75 percent completion MCQs in kinetics. Such graphical interpretation is frequently included in 50 percent and 75 percent completion MCQs in kinetics, making visualization skills equally important.
In addition to first-order reactions, zero-order and second-order reactions may also involve percentage completion problems. However, the calculations are slightly different because the rate laws for these reactions follow different mathematical forms. Aspirants preparing for exams therefore practice 50 percent and 75 percent completion MCQs in kinetics to become familiar with how each reaction order behaves.
Another benefit of solving 50 percent and 75 percent completion MCQs in kinetics is that it strengthens numerical problem-solving skills. Many examination questions involve converting percentage completion into remaining fractions of reactants and then applying kinetic equations. Repeated exposure to these 50 percent and 75 percent completion MCQs in kinetics problems helps aspirants perform calculations quickly and accurately during exams.
Furthermore, these types of problems help aspirants connect theoretical chemical kinetics with real-world chemical processes. In industrial chemistry, pharmaceutical manufacturing, and environmental chemistry, understanding how long a reaction takes to reach certain levels of completion is very important. Practicing 50 percent and 75 percent completion MCQs in kinetics therefore prepares aspirants not only for exams but also for practical scientific applications.
Regular practice with 50 percent and 75 percent completion MCQs in kinetics helps aspirants understand the relationship between reaction progress and time. By solving 50 percent and 75 percent completion MCQs in kinetics, aspirants strengthen their conceptual clarity and improve problem-solving speed. As a result, 50 percent and 75 percent completion MCQs in kinetics becomes essential practice tool for aspirants preparing for competitive exams like NEET, JEE & CUET.
Aspirants also learn how reaction rate constants influence the time required for certain percentages of completion. When the rate constant is large, the reaction proceeds faster, and the time required to reach 50 percent or 75 percent completion MCQs in kinetics becomes shorter. This relationship is often explored through 50 percent and 75 percent completion MCQs in kinetics, helping aspirants understand how reaction speed depends on kinetic parameters.
Another key aspect of mastering chemical kinetics is learning how logarithmic calculations are used in solving percentage completion problems like 50 percent and 75 percent completion MCQs in kinetics. Since many first-order reaction equations involve natural logarithms, aspirants must be comfortable manipulating these expressions. Regular practice with 50 percent and 75 percent completion MCQs in kinetics ensures that aspirants develop confidence in using these mathematical tools.
50 percent and 75 percent completion MCQs in kinetics:
Half-life period of a first order reaction, A → product is 6.93 h. What is the value of rate constant?
A. 1.596 h⁻¹
B. 0.1 h⁻¹
C. 4.802 h⁻¹
D. 0.693 h⁻¹
Answer: B
Q2.
The relationship between rate constant and half-life period of zero order reaction is given by
A. t1/2=[A0]/2kt_{1/2} = [A_0]/2k
B. t1/2=0.693/kt_{1/2} = 0.693/k
C. t1/2=[A0]/2kt_{1/2} = [A_0]/2k
D. None of these
Answer: C
Q3.
When initial concentration of a reactant is doubled in a reaction, its half-life period is not affected. The order of the reaction is
A. Second
B. Zero
C. More than zero but less than first
D. First
Answer: D
Q4.
The half-life period of second order reaction is proportional to
A. a1a^1
B. a0a^0
C. a−2a^{-2}
D. a−1a^{-1}
Answer: D
Q5.
The half-life period of a first order chemical reaction is 6.93 minutes. The time required for completion of 99% reaction will be
A. 23.03 minutes
B. 46.06 minutes
C. 26.06 minutes
D. 230.3 minutes
Answer: B
Q6.
For a first order reaction at 27°C, the ratio of time required for 75% completion to 25% completion of reaction is
A. 3
B. 2.303
C. 4.8
D. 0.477
Answer: C
Q7.
In a first order reaction, concentration decreases from 0.6 M to 0.3 M in 15 minutes. Time required for change from 0.1 M to 0.025 M is
A. 12 min
B. 6 min
C. 18 min
D. 30 min
Answer: D
Q8.
When Zn dust is added to a solution of MgCl₂
A. Magnesium is precipitated
B. Zinc dissolves in solution
C. Zinc chloride is formed
D. No reaction will take place
Answer: D
Q9.
The fraction of element disintegrated after 4 half-lives in percentage is
A. 75%
B. 87.5%
C. 93.75%
D. 92.5%
Answer: C
Q10.
If 0.4 Curie is the activity of 1 g radioactive sample (atomic mass 226), the half-life period is
A. 1.23×10111.23 \times 10^{11} s
B. 1.8×10111.8 \times 10^{11} s
C. 1.23×10101.23 \times 10^{10} s
D. 1.8×10101.8 \times 10^{10} s
Answer: A
Q11.
6.25% of radioactive substance is left after 480 minutes. Half-life period is
A. 60 min
B. 90 min
C. 150 min
D. 120 min
Answer: D
Q12.
A reaction is 50% complete in 2 hours and 75% complete in 4 hours. Order of reaction is
A. 1
B. 2
C. 3
D. 4
Answer: A
Q13.
Relation between half-life time and initial concentration for (n−1) order reaction
A. t1/2∝[R0]t_{1/2} \propto [R_0]
B. t1/2∝[R0]2−nt_{1/2} \propto [R_0]^{2-n}
C. t1/2∝[R0]n+1t_{1/2} \propto [R_0]^{n+1}
D. t1/2∝[R0]n−2t_{1/2} \propto [R_0]^{n-2}
Answer: B
Q14.
For a first-order reaction initial concentration is 0.05 M. After 45 min it decreases by 0.015 M. Half-life is
A. 42.72 min
B. 25.90 min
C. 87.42 min
D. 77.20 min
Answer: C
Q15.
Formula for half-life of zero order reaction
A. 0.693/K0.693/K
B. [R0]/2K[R_0]/2K
C. 2K/[R0]2K/[R_0]
D. K/[R0]K/[R_0]
Answer: B
Q16.
For n order reaction, half-life proportionality is
A. 1/an−11/a^{n-1}
B. an+1a^{n+1}
C. a1a^1
D. a0a^0
Answer: A
Q17.
In a first-order reaction A → B, half-life is
A. ln2/kln2/k
B. 0.693/0.50.693/0.5
C. log2/2klog2/2k
D. log2/6klog2/6k
Answer: A
Q18.
Half-life of reaction is 1 hour. If zero order, time required for concentration to change from 0.50 to 0.25 mol L⁻¹ is
A. 0.5 h
B. 1 h
C. 0.25 h
D. 4 h
Answer: C
Q19.
For first order reaction k=3.01×10−3s−1k = 3.01 \times 10^{-3} s^{-1}, time required for half decomposition is
A. 230.3 s
B. 2303 s
C. 23.03 s
D. 23030 s
Answer: A
Q20.
For second order reaction 2A→products2A → products, k=10−4Lmol−1min−1k = 10^{-4} L mol^{-1} min^{-1} and concentration 10−210^{-2}. Half-life is
A. 1000 min
B. 10610^6 min
C. 10 min
D. 10000 min
Answer: B
Q21.
For zero order reaction, half-life relation with initial concentration is
A. t1/2∝1/at_{1/2} \propto 1/a
B. t1/2∝at_{1/2} \propto a
C. t1/2∝1/a2t_{1/2} \propto 1/a^2
D. t1/2∝a2t_{1/2} \propto a^2
Answer: B
Q22.
If 75% of first order reaction completes in 8 min, time for 50% completion is
A. 2 min
B. 4 min
C. 12 min
D. 1 min
Answer: B
Q23.
Half-life is 6 min. Initial amount 32 g. Amount after 18 min
A. 4 g
B. 8 g
C. 16 g
D. 2 g
Answer: A
Q24.
For first order reaction, rate at concentration 0.01 M is 2.0×10−52.0 \times 10^{-5}. Half-life is
A. 220 s
B. 30 s
C. 300 s
D. 347 s
Answer: D
Q25.
Half-life of first order reaction with k=1.7×10−5s−1k = 1.7 \times 10^{-5} s^{-1}
A. 12.1 h
B. 9.7 h
C. 11.3 h
D. 1.8 h
Answer: C
Q26.
For reaction A(g)→P+2Q+RA(g)→P+2Q+R with half-life 69.3 s and initial pressure 0.4 atm, pressure after 230 s is
A. 1.15 atm
B. 1.32 atm
C. 1.22 atm
D. 1.12 atm
Answer: D
Q27.
Two-third life of first order reaction with k=5.48×10−14k = 5.48 \times 10^{-14}
A. 0.201×1013s0.201 \times 10^{13} s
B. 2.01×1013s2.01 \times 10^{13} s
C. 2.01×1015s2.01 \times 10^{15} s
D. None
Answer: B
Q28.
For a first order reaction, half-life
A. Depends on square of concentration
B. Depends on concentration
C. Depends on square root of concentration
D. Independent of concentration
Answer: D
Q29.
Half-life of enzyme catalyzed reaction = 138 s. Time required for concentration change from 1.28 mg/L to 0.04 mg/L
A. 414 s
B. 552 s
C. 690 s
D. 276 s
Answer: C
Q30.
Half-life of first order reaction = 1386 s. Rate constant is
A. 5.0×10−3s−15.0 \times 10^{-3} s^{-1}
B. 0.5×10−3s−10.5 \times 10^{-3} s^{-1}
C. 5.5×10−3s−15.5 \times 10^{-3} s^{-1}
D. 5.0×10−5s−15.0 \times 10^{-5} s^{-1}
Answer: B
Conclusion on 50 percent and 75 percent completion MCQs in kinetics
In conclusion, percentage completion problems are an essential part of chemical kinetics. They help aspirants understand how reactions progress over time and how mathematical relationships describe reaction rates. By practicing 50 percent and 75 percent completion MCQs in kinetics, aspirants gain valuable experience in applying kinetic equations, interpreting graphs, and solving numerical problems. Consistent practice with 50 percent and 75 percent completion MCQs in kinetics ultimately strengthens conceptual understanding and prepares aspirants for success in chemistry examinations.
