- Keneitsino Lydia
- March 10, 2026
Master 30 Temperature Effect on Reaction Rate MCQs – Powerful Practice for NEET, JEE & CUET
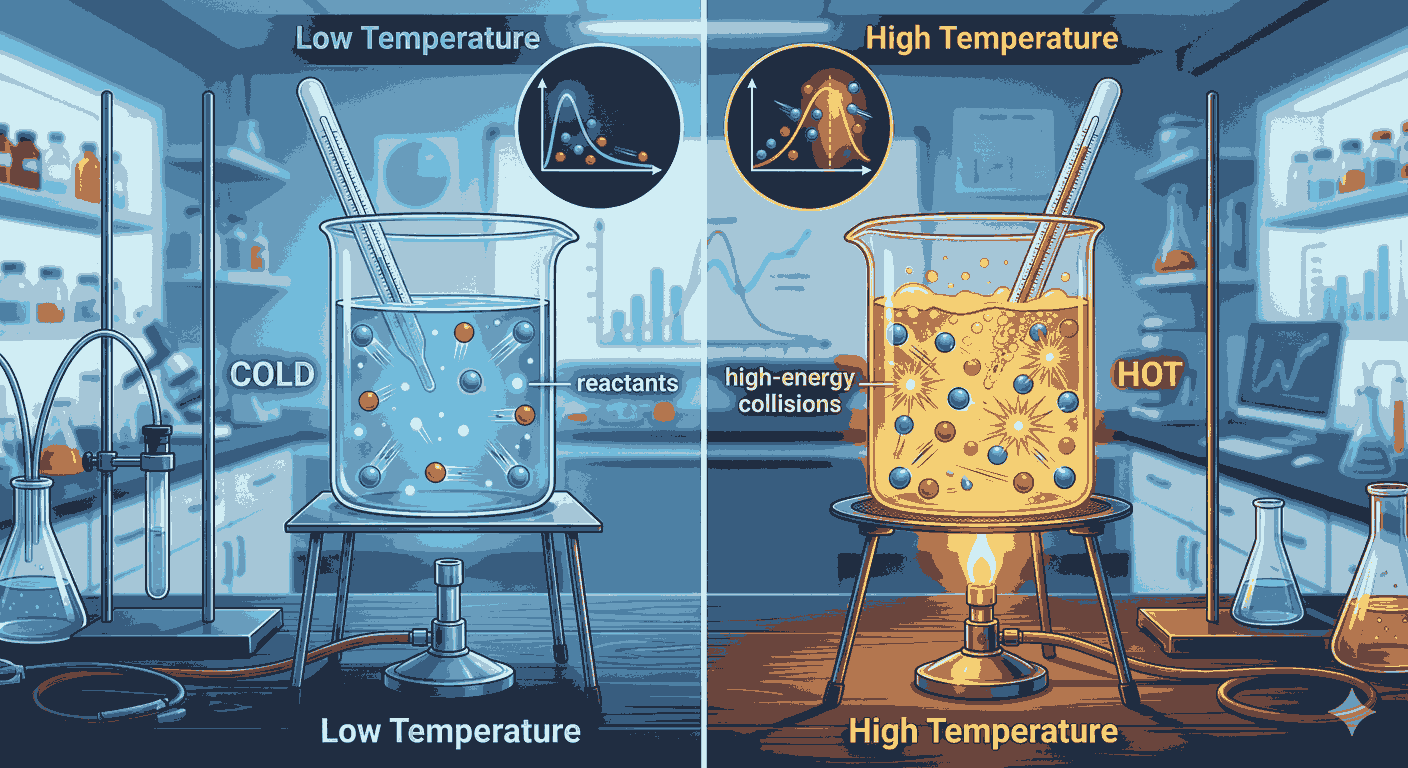
In temperature effect on reaction rate mcqs, Temperature is one of the most important factors affecting the rate of a chemical reaction. In chemical kinetics, even a small change in temperature can significantly influence how fast reactants convert into products. Because of its importance, many aspirants frequently practice Temperature effect on reaction rate mcqs to understand how temperature influences reaction speed and reaction mechanisms. Studying Temperature effect on reaction rate mcqs helps build strong conceptual knowledge of kinetic theory and reaction dynamics.
In Temperature effect on reaction rate mcqs, when the temperature of a reaction system increases, the kinetic energy of molecules also increases. This means that molecules move faster and collide more frequently with each other. However, not all collisions result in a reaction. Only those collisions that possess sufficient energy to overcome the activation energy barrier lead to product formation. This concept is often emphasized in Temperature effect on reaction rate mcqs, where aspirants analyze how temperature changes influence the probability of effective collisions.
The relationship between temperature and reaction rate is explained mathematically through the Arrhenius equation. This equation shows that the rate constant of a reaction increases exponentially with temperature. As a result, even a small rise in temperature can cause a significant increase in the reaction rate. Many Temperature effect on reaction rate mcqs involve applying the Arrhenius equation to calculate how rate constants change with temperature. Practicing Temperature effect on reaction rate mcqs helps aspirants become comfortable using these equations in numerical problems.
Another important concept related to temperature effect on reaction rate mcqs is activation energy. Activation energy is the minimum energy required for reactant molecules to transform into products. At higher temperatures, a larger fraction of molecules possesses energy greater than the activation energy, allowing more reactions to occur. This idea frequently appears in Temperature effect on reaction rate mcqs, helping aspirants understand the molecular explanation behind temperature-dependent reaction rates.
Graphical representation also plays a key role in understanding temperature effect on reaction rate mcqs. The Arrhenius plot, which is a graph of the logarithm of the rate constant versus the inverse of temperature, provides a linear relationship that helps determine activation energy. Many Temperature effect on reaction rate mcqs ask aspirants to interpret such graphs and calculate activation energies from the slope. Learning these graphical methods through Temperature effect on reaction rate mcqs strengthens analytical skills in chemical kinetics.
In temperature effect on reaction rate mcqs, many reactions, increasing the temperature by 10°C approximately doubles the reaction rate. This empirical observation is commonly used to estimate how reaction speed changes with temperature. Such relationships are often tested in Temperature effect on reaction rate mcqs, especially in board examinations and competitive entrance tests. By solving Temperature effect on reaction rate mcqs, aspirants develop a deeper understanding of how temperature influences reaction kinetics.
Temperature also affects reactions differently depending on whether a catalyst is present. Catalysts lower the activation energy required for the reaction, making the reaction faster even at lower temperatures. However, increasing temperature can still further accelerate catalyzed reactions. Many Temperature effect on reaction rate mcqs explore how catalysts and temperature together influence reaction rates, helping aspirants connect different concepts within chemical kinetics.
In industrial chemistry and biological systems, controlling temperature is essential for maintaining optimal reaction rates. Chemical industries often regulate temperature carefully to achieve maximum efficiency and product yield. Similarly, enzymes in biological systems operate within specific temperature ranges to function effectively. These practical applications are often highlighted in Temperature effect on reaction rate mcqs, showing the real-world importance of temperature in chemical reactions.
Temperature Effect on Reaction Rate MCQs:
1. Activation energy from rate constants
Q1. For the reaction of H₂ with I₂, the rate constant is 2.5×10−4 dm3 mol−1 s−12.5 \times 10^{-4}\ \mathrm{dm^3\,mol^{-1}\,s^{-1}} at 327°C and 1.0 dm3 mol−1 s−11.0\ \mathrm{dm^3\,mol^{-1}\,s^{-1}} at 527°C. The activation energy for the reaction is:
A. 59 kJ mol⁻¹
B. 79 kJ mol⁻¹
C. 89 kJ mol⁻¹
D. 166 kJ mol⁻¹
Answer: D
2. Temperature Rise Effect
Q2. For many reactions, the rate of reaction approximately doubles when the temperature increases by:
A. 5°C
B. 10°C
C. 15°C
D. 20°C
Answer: B
3. Gradient of lnk\ln k vs 1/RT1/RT
Q3. If a reaction follows the Arrhenius equation, the plot of lnk\ln k versus 1/RT1/RT gives a straight line with gradient (−y)(-y). The energy required to activate the reactant is:
A. EaE_a unit
B. RR unit
C. yRyR unit
D. yy unit
Answer: D
4. Wrong statement
Q4. Which of the following is the wrong statement?
A. k=Ak = A if Ea=0E_a = 0
B. e−Ea/RTe^{-E_a/RT} gives the fraction of molecules activated at a given temperature
C. Plot of lnk\ln k versus 1/T1/T is a straight line
D. Presence of catalyst will not alter the value of EaE_a
Answer: D
5. Zero activation energy
Q5. The activation energy of a reaction is zero. Its rate constant at 280 K is 1.6×10−6 s−11.6 \times 10^{-6}\ \mathrm{s^{-1}}. The rate constant at 300 K is:
A. 3.2×10−4 s−13.2 \times 10^{-4}\ \mathrm{s^{-1}}
B. 1.6×10−6 s−11.6 \times 10^{-6}\ \mathrm{s^{-1}}
C. Zero
D. 1.6×10−4 s−11.6 \times 10^{-4}\ \mathrm{s^{-1}}
Answer: B
6. Another zero activation energy question
Q6. For a reaction, Ea=0E_a = 0, and the rate constant at 200 K is 1.6×10−6 s−11.6 \times 10^{-6}\ \mathrm{s^{-1}}. The rate constant at 400 K will be:
A. 1.6×104 s−11.6 \times 10^{4}\ \mathrm{s^{-1}}
B. 1.6×10−6 s−11.6 \times 10^{-6}\ \mathrm{s^{-1}}
C. 1.6×10−4 s−11.6 \times 10^{-4}\ \mathrm{s^{-1}}
D. 3.2×10−4 s−13.2 \times 10^{-4}\ \mathrm{s^{-1}}
Answer: B
7. Doubling of rate constant
Q7. The rate constant is doubled when temperature increases from 27°C to 37°C. The activation energy is:
A. 34 kJ
B. 54 kJ
C. 50 kJ
D. 100 kJ
Answer: B
8. Activation energy on heating from 300 K to 400 K
Q8. What is the activation energy for a reaction if its rate constant doubles when temperature is raised from 300 K to 400 K?
A. 68.8 kJ mol⁻¹
B. 6.88 kJ mol⁻¹
C. 34.4 kJ mol⁻¹
D. 3.44 kJ mol⁻¹
Answer: B
9. Arrhenius Plot
Q9. A plot of In k versus 1/T according to Arrhenius equation gives:
A. Parabola
B. Straight line
C. Hyperbola
D. Circle
Answer: B
10. Expression for activation energy
Q10. The rate constant of a reaction is 1.5×10−31.5 \times 10^{-3} at 25°C and 2.1×10−22.1 \times 10^{-2} at 60°C. The correct expression for activation energy is:
A. 35333Rln(2.1×10−21.5×10−2)\frac{35}{333}R\ln\left(\frac{2.1\times10^{-2}}{1.5\times10^{-2}}\right)
B. 298×33335Rln(211.5)\frac{298\times333}{35}R\ln\left(\frac{21}{1.5}\right)
C. 298×33335Rln(2.1)\frac{298\times333}{35}R\ln(2.1)
D. 298×33335Rln(2.11.5)\frac{298\times333}{35}R\ln\left(\frac{2.1}{1.5}\right)
Answer: B
11. Arrhenius plot
Q11. Under normal circumstances, the Arrhenius plot is obtained by plotting:
A. log(1/k)\log(1/k) versus 1/T1/T
B. logk\log k versus TT
C. logk\log k versus 1/T1/T
D. log(1/k)\log(1/k) versus TT
Answer: C
12. Half-life problem using Arrhenius equation
Q12. For a certain reaction, A=4×1013 s−1A = 4 \times 10^{13}\ \mathrm{s^{-1}} and Ea=98.6 kJ mol−1E_a = 98.6\ \mathrm{kJ\,mol^{-1}}. If the reaction is first order, the temperature at which the half-life is 10 minutes is:
A. 325.60 K
B. 311.35 K
C. 300 K
D. 520.05 K
Answer: B
13. Temperature where two rate constants are equal
Q13. For the gaseous reactions
A→Product, k1=1015e−2000/TA \to \text{Product},\ k_1 = 10^{15}e^{-2000/T}
C→D, k2=1014e−1000/TC \to D,\ k_2 = 10^{14}e^{-1000/T}
The temperature at which k1=k2k_1 = k_2 is:
A. 2000 K
B. 868.8 K
C. 434.2 K
D. 1000 K
Answer: C
14. Find EaE_a and kk
Q14. For the equation lnk=−2576T+12.1\ln k = -\frac{2576}{T} + 12.1, the value of EaE_a and kk at 27°C are:
A. Ea=21.416 kJE_a = 21.416\ \mathrm{kJ}, k=0.335×102 min−1k = 0.335 \times 10^2\ \mathrm{min^{-1}}
B. Ea=11.4 kJE_a = 11.4\ \mathrm{kJ}, k=0.335×107 min−1k = 0.335 \times 10^7\ \mathrm{min^{-1}}
C. Ea=21.4 kJE_a = 21.4\ \mathrm{kJ}, k=0.3×107 min−1k = 0.3 \times 10^7\ \mathrm{min^{-1}}
D. Ea=21.416 kJE_a = 21.416\ \mathrm{kJ}, k=0.335×102 mink = 0.335 \times 10^2\ \mathrm{min}
Answer: A
15. Arrhenius equation form
Q15. Which of the following represents the Arrhenius equation?
A. k=AeEa/RTk = Ae^{E_a/RT}
B. k=AeRT/Eak = Ae^{RT/E_a}
C. k=AeEa/RTk = \frac{A}{e^{E_a/RT}}
D. None of these
Answer: C
16. Backward activation energy
Q16. For the reaction A→BA \to B, ΔH=20 kJ mol−1\Delta H = 20\ \mathrm{kJ\,mol^{-1}} and activation energy of forward reaction is 85 kJ mol⁻¹. The activation energy of backward reaction is:
A. 105 kJ mol⁻¹
B. 65 kJ mol⁻¹
C. 45 kJ mol⁻¹
D. 75 kJ mol⁻¹
Answer: B
17. Endothermic reaction and minimum activation energy
Q17. For an endothermic reaction, the minimum value of activation energy for the forward reaction will be:
A. Less than ΔH\Delta H
B. More than ΔH\Delta H
C. Equal to ΔH\Delta H
D. Zero
Answer: B
18. Correct logk\log k vs 1/T1/T plot
Q18. The correct graph for logk\log k versus 1/T1/T used to calculate EaE_a is:
A. Option 1
B. Option 2
C. Option 3
D. Option 4
Answer: B
19. Intercept of lnK\ln K vs 1/T1/T
Q19. A plot of lnK\ln K against 1/T1/T is expected to be a straight line with intercept on the ordinate equal to:
A. ΔS∘/R\Delta S^\circ/R
B. ΔH∘/R\Delta H^\circ/R
C. 2.303R2.303R
D. RΔS∘R\Delta S^\circ
Answer: A
20. Slope of Arrhenius graph
Q20. The slope of the graph drawn between lnk\ln k and 1/T1/T gives:
A. Ea/RE_a/R
B. −Ea/R-E_a/R
C. EaE_a
D. −Ea-E_a
Answer: B
21. Backward activation energy from enthalpy and forward EaE_a
Q21. The formation of H₂O₂ in the upper atmosphere follows a mechanism with ΔH=72 kJ mol−1\Delta H = 72\ \mathrm{kJ\,mol^{-1}} and Ea=77 kJ mol−1E_a = 77\ \mathrm{kJ\,mol^{-1}}. The activation energy for the backward reaction is:
A. −149 kJ
B. 149 kJ
C. −5 kJ
D. 5 kJ
Answer: D
22. Catalyst and temperature
Q22. A hydrogenation reaction is carried out at 500 K. In the presence of a catalyst, the same rate is obtained at 400 K. If the catalyst lowers the activation barrier by 40 kJ mol⁻¹, the activation energy of the uncatalyzed reaction is:
A. 10 kJ mol⁻¹
B. 200 kJ mol⁻¹
C. 300 kJ mol⁻¹
D. 175 kJ mol⁻¹
Answer: B
23. Effect of catalyst
Q23. Addition of a catalyst during a reaction alters:
A. Enthalpy
B. Activation energy
C. Entropy
D. Internal energy
Answer: B
24. Arrhenius Equation
Q24. The relationship between rate constant and temperature is given by:
A. Nernst equation
B. Arrhenius equation
C. Gibbs equation
D. Van’t Hoff equation
Answer: B
25. Unit of activation energy
Q25. The unit of activation energy is:
A. s⁻¹
B. K⁻¹ mol⁻¹
C. s
D. J mol⁻¹
Answer: D
26. Factor by which catalyst increases rate
Q26. For a reaction at 300 K, the uncatalyzed EaE_a is 76 kJ mol⁻¹ and catalyzed EaE_a is 57 kJ mol⁻¹. The factor by which the catalyzed reaction rate increases is:
A. 21
B. 2100
C. 2000
D. 1200
Answer: C
27. Backward activation energy for endothermic process
Q27. For an endothermic process, forward activation energy is 90 kJ and ΔH=50\Delta H = 50 kJ. The backward activation energy is:
A. 40 kJ
B. 90 kJ
C. 140 kJ
D. 50 kJ
Answer: A
28. Graph used to determine activation energy
Q28. Activation energy can be determined from the slope of which graph?
A. lnk\ln k vs 1/T1/T
B. lnk\ln k vs TT
C. logk\log k vs TT
D. TlnkT\ln k vs TT
Answer: A
29. Wrong statement from energy profile
Q29. For a reversible reaction A⇌BA \rightleftharpoons B, which statement is wrong from the energy profile diagram?
A. Activation energy of forward reaction is greater than backward reaction
B. Forward reaction is endothermic
C. Threshold energy is less than activation energy
D. Forward activation energy equals heat of reaction plus backward activation energy
Answer: C
30. Zero activation energy again
Q30. For a reaction, Ea=0E_a = 0, and k=4.2×105 s−1k = 4.2 \times 10^5\ \mathrm{s^{-1}} at 300 K. The value of kk at 310 K will be:
A. 4.2×105 s−14.2 \times 10^5\ \mathrm{s^{-1}}
B. 8.4×106 s−18.4 \times 10^6\ \mathrm{s^{-1}}
C. 4.2×107 s−14.2 \times 10^7\ \mathrm{s^{-1}}
D. Unpredictable
Answer: A
Conclusion on Temperature Effect On Reaction Rate MCQs
In conclusion, temperature effect on reaction rate mcqs plays a crucial role in determining the speed of chemical reactions. By practicing Temperature effect on reaction rate mcqs, aspirants can strengthen their understanding of activation energy, Arrhenius equations, and molecular collision theory. Consistent practice with Temperature effect on reaction rate mcqs improves problem-solving skills and prepares aspirants for exams where reaction rate concepts are frequently tested.
