- Keneitsino Lydia
- March 17, 2026
Tyndall Effect Objective Questions Class 12 Chemistry: Powerful MCQs to Boost Your Score
The study of colloids in Class 12 chemistry introduces several fascinating phenomena, and one of the most visually striking among them is the Tyndall effect. This concept is essential for understanding how light interacts with colloidal particles and is frequently tested in board exams as well as competitive entrance tests. Because of its importance, many aspirants focus on mastering the concept through Tyndall Effect Objective Questions Class 12 Chemistry. Regular practice of Tyndall Effect Objective Questions Class 12 Chemistry helps in strengthening both conceptual clarity and problem-solving ability.
The Tyndall effect refers to the scattering of light by colloidal particles present in a dispersion medium. When a beam of light passes through a colloidal solution, the path of light becomes visible due to scattering. This phenomenon does not occur in true solutions because the particle size is too small to scatter light effectively. Understanding this distinction is crucial, and it is often emphasized through Tyndall Effect Objective Questions Class 12 Chemistry. By solving Tyndall Effect Objective Questions Class 12 Chemistry, aspirants can easily differentiate between true solutions, colloids, and suspensions.
The effectiveness of the Tyndall effect depends on two main factors: the size of the dispersed particles and the difference in refractive index between the dispersed phase and the dispersion medium. When the particle size is comparable to the wavelength of light, scattering occurs efficiently. These concepts are commonly tested in Tyndall Effect Objective Questions Class 12 Chemistry, making it important for aspirants to revise them thoroughly. Practicing Tyndall Effect Objective Questions Class 12 Chemistry ensures that aspirants understand how physical parameters influence light scattering.
Another important aspect of the Tyndall effect is its practical application. It is used to detect the presence of colloidal particles in a solution. For example, when sunlight enters a dusty room, the particles scatter light, making the beam visible. Similarly, in laboratories, the Tyndall effect helps distinguish between colloidal solutions and true solutions. These real-life applications are frequently included in Tyndall Effect Objective Questions Class 12 Chemistry, allowing aspirants to connect theoretical knowledge with everyday observations. Reviewing Tyndall Effect Objective Questions Class 12 Chemistry enhances both conceptual and practical understanding.
It is also important to note that not all systems show the Tyndall effect. True solutions do not exhibit this phenomenon because their particles are too small to scatter light. On the other hand, suspensions show scattering but often settle down over time. Colloids, however, remain stable and continue to scatter light, which makes the Tyndall effect a characteristic feature of colloidal systems. These distinctions are often tested through Tyndall Effect Objective Questions Class 12 Chemistry, making them essential for exam preparation. Practicing Tyndall Effect Objective Questions Class 12 Chemistry helps aspirants avoid common mistakes.
The intensity of the Tyndall effect can vary depending on the concentration of the colloidal particles. Higher concentration leads to greater scattering of light, making the path of light more visible. Additionally, the wavelength of light used can also affect the scattering intensity. These variations are frequently explored in Tyndall Effect Objective Questions Class 12 Chemistry, helping aspirants develop a deeper understanding of the concept. By working through Tyndall Effect Objective Questions Class 12 Chemistry, aspirants can confidently handle application-based questions.
The Tyndall effect also plays a role in environmental and industrial processes. For instance, it is used in air pollution studies to detect particulate matter in the atmosphere. In industries, it helps in analyzing colloidal suspensions and ensuring product quality. Such applications are often included in Tyndall Effect Objective Questions Class 12 Chemistry, providing a broader perspective on the topic. Practicing Tyndall Effect Objective Questions Class 12 Chemistry allows aspirants to appreciate the relevance of chemistry in real-world scenarios.
Tyndall Effect Objective Questions Class 12 Chemistry:
Q1.
Which of the following is a lyophobic colloid?
A. Sulphur
B. Starch
C. Gum Arabic
D. Gelatin
Answer: A
Q2.
Curd belongs to which type of colloid?
A. Gel
B. Sol
C. Emulsion
D. Solid form
Answer: A
Q3.
Which substance acts as a negative catalyst in decomposition of H₂O₂?
A. Platinum
B. Alcohol
C. Glycerol
D. MnO₂
Answer: C
Q4.
The Tyndall effect is not observed in:
A. Emulsion
B. True solution
C. Colloidal solution
D. Solid sol
Answer: B
Q5.
In stopping bleeding using potash alum, alum acts as:
A. Germicide
B. Coagulating agent
C. Fungicide
D. Disinfectant
Answer: B
Q6.
Kala-azar disease is treated using:
A. Colloidal antimony
B. Milk of magnesia
C. Argyrol
D. Colloidal gold
Answer: A
Q7.
Which statement is incorrect for liquids?
A. Surface tension decreases with temperature
B. Viscosity decreases with decrease in temperature
C. Vapour pressure increases with temperature
D. Droplets flatten due to gravity
Answer: B
Q8.
Identify the false statement:
A. Colloidal sols are homogeneous
B. Colloids carry charge
C. Colloids show Tyndall effect
D. Particle size is 10–1000 Å
Answer: A
Q9.
Micelles are:
A. Adsorbed catalyst
B. Ideal solution
C. Associated colloids
D. Associated colloids
Answer: D
Q10.
Dissolution of sulphur (S₈) forms:
A. Associated colloid
B. Micelle
C. Multimolecular colloid
D. Macromolecular colloid
Answer: C
Q11.
Solid dispersed in liquid is classified as:
A. Solid sol
B. Gel
C. Emulsion
D. Sol
Answer: D
Q12.
Which phenomenon is related to colloids?
A. Tyndall effect
B. Fajan’s rule
C. Le Chatelier principle
D. Aufbau principle
Answer: A
Q13.
Which statement is incorrect?
A. Milk is an emulsion
B. Gold sol is lyophilic
C. Physical adsorption decreases with temperature
D. Chemisorption is monolayer
Answer: B
Q14.
Greater protective power corresponds to:
A. Higher gold number
B. Lower gold number
C. Constant value
D. None
Answer: B
Q15.
Negative adsorption occurs in:
A. Acetic acid + charcoal
B. Dilute KCl + charcoal
C. Concentrated KCl + charcoal
D. H₂ + charcoal
Answer: B
Q16.
Peptisation involves:
A. Precipitation
B. Disintegration of aggregates
C. Evaporation
D. Molecular impact
Answer: B
Q17.
Most effective electrolyte for coagulating As₂S₃ sol:
A. KCl
B. AlCl₃
C. MgSO₄
D. K₃Fe(CN)₆
Answer: B
Q18.
Milk changes after digestion into:
A. Cellulose
B. Fructose
C. Glucose
D. Lactose
Answer: C
Q19.
The continuous phase contains the dispersed phase throughout. Example:
A. Water in milk
B. Fat in milk
C. Water droplets in mist
D. Oil in water
Answer: A
Q20.
The size of particles in suspension, colloidal solution, and true solution varies as:
A. Suspension > Colloidal > True solution
B. True solution > Suspension > Colloidal
C. Suspension > Colloidal = True solution
D. None of these
Answer: A
Q21.
Size of colloidal particles is:
A. 1 nm to 100 nm
B. 1 mμ to 100 mμ
C. 0.05 μm to 0.1 μm
D. 25 μm to 30 μm
Answer: B
Q22.
The diameter of colloidal particles ranges from:
A. 1 Å to 10 Å
B. 10 Å to 200 Å
C. 10 Å to 1000 Å
D. 100 Å to 2000 Å
Answer: C
Q23.
Silver sol used as an eye lotion is:
A. Milk of magnesia
B. Argentite
C. Argyrol
D. Silver chloride
Answer: C
Q24.
The Tyndall effect is due to :
A.Reflection of light
B.Refraction of light
C.Scattering of light by colloidal particles
D. Absorption of light
Answer: C
Q25.
Gel is a colloidal solution of:
A. Solid in liquid
B. Liquid in solid
C. Solid in solid
D. Solid in gas
Answer: B
Q26.
SO₂ + 2H₂S → 3S + 2H₂O represents preparation of sulphur sol by:
A. Hydrolysis
B. Oxidation
C. Reduction
D. Double decomposition
Answer: B
Q27.
The physical states of dispersed phase and dispersion medium in pesticide spray are:
A. Solid, gas
B. Gas, liquid
C. Liquid, gas
D. Liquid, solid
Answer: C
Q28.
As₂O₃ + 3H₂S → As₂S₃ (sol) + 3H₂O is an example of:
A. Oxidation
B. Hydrolysis
C. Reduction
D. Double decomposition
Answer: D
Q29.
The charge on colloidal particles when FeCl₃ is added to excess hot water is:
A. Positive
B. Negative
C. Neutral
D. Sometimes positive and sometimes negative
Answer: A
Q30.
Which one of the following statements is FALSE for hydrophilic sols?
A. They do not require electrolytes for stability
B. These sols are reversible in nature
C. Their viscosity is of the order of that of H₂O
D. The sols cannot be easily coagulated
Answer: C
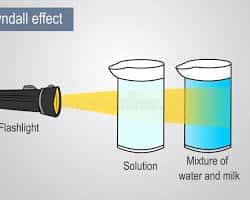
Conclusion on Tyndall Effect Objective Questions Class 12 Chemistry:
In conclusion, the Tyndall effect is a fundamental concept in surface chemistry that helps in identifying and studying colloidal systems. Its dependence on particle size, refractive index, and concentration makes it an important topic for exams. A clear understanding of this phenomenon can be achieved through consistent practice and revision. Using Tyndall Effect Objective Questions Class 12 Chemistry as a study tool enables aspirants to strengthen their knowledge and improve their performance. Ultimately, mastering Tyndall Effect Objective Questions Class 12 Chemistry ensures better accuracy and confidence in solving chemistry questions.
